Case
Dr. Swan, a young neurosurgeon, felt a nearly palpable anxiety in the air as he entered the examination room to see Mrs. Jones.
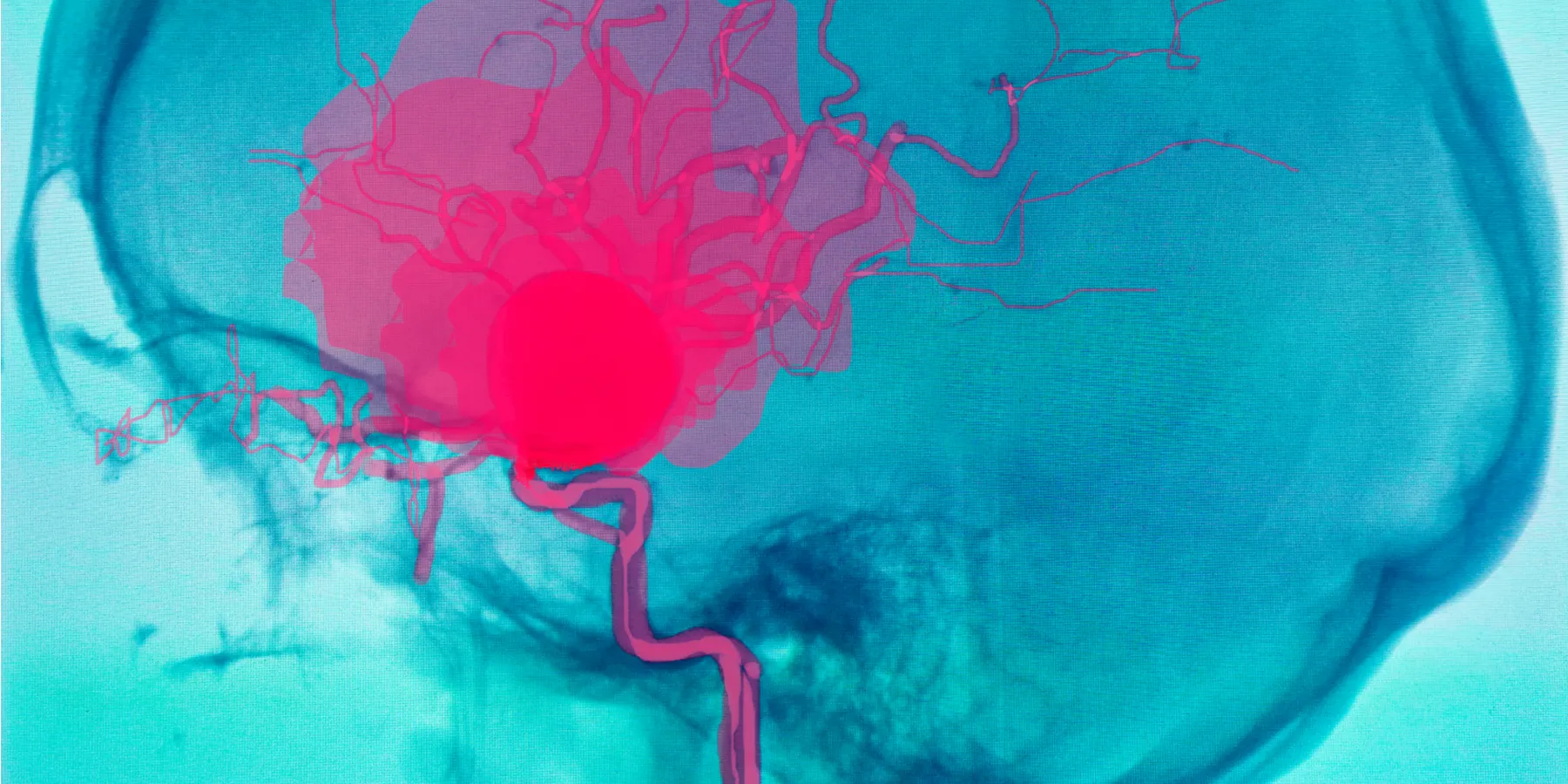
Mrs. Jones had been diagnosed with an unruptured cerebral aneurysm and referred to Dr. Swan. Her primary care physician had ordered an MRI scan of her head to work up her recurrent headaches, which were, ultimately, diagnosed as tension headaches. However, her workup incidentally revealed a moderate-sized aneurysm of the right posterior communicating artery.
In her initial visit, Dr. Swan had counseled Mrs. Jones that her aneurysm carried a roughly 1 percent annual risk of rupture and that a rupture carried a roughly two-thirds chance of neurologic disability or death. After conferring with his partners in interventional neuroradiology, Dr. Swan and his team recommended craniotomy on the basis of the characteristics of her aneurysm. In response to Mrs. Jones’s questions about the danger of the surgery, he said, “If everything goes well, you could be out of the hospital three days after your craniotomy and the aneurysm will be gone. However, all surgeries have risk and this one is a complicated, delicate procedure. In my experience, you would have a roughly 15 percent risk of suffering a stroke or neurological deficit from surgery. The risk of wound infection, which could potentially result in a washout surgery, is about 5-8 percent. The surgery could also result in death, although this is rare, less than 1 percent of the time. Of course, every patient is different and no one has ever done a study on you, but this is what our experience tells us.” Momentarily overwhelmed, Mrs. Jones asked to postpone treatment and think it over.
Dr. Swan reassured Mrs. Jones that, although hers was not an emergency, delaying treatment would result in no benefit and would yet expose her to a continued risk of neurologic catastrophe.
Several weeks later, Mrs. Jones returned to Dr. Swan’s clinic for a followup visit. Dr. Swan scrutinized the new angiogram study he had ordered as part of her preoperative workup. To his concern, the aneurysm appeared to have subtly increased in size. Mrs. Jones, however, was not ready to provide consent for a craniotomy. She told Dr. Swan: “I have been thinking about all those risks you mentioned, like disability from stroke, and I wouldn’t want my children to see me like that. I’m a gambler. At my age, I might just take my chances with a tiny annual risk rather than the known risks of going under the knife.”
Dr. Swan was taken aback. Although he had recommended surgery, he was determined to respect his patient’s wishes. Mrs. Jones appeared to understand the details of her medical condition and the risks and benefits of Dr. Swan’s treatment recommendation. However, she decided not to pursue treatment because of her age and her own self-described “gambler” disposition in the face of personal risk.
Dr. Swan was reminded of a popular magazine article he had seen, describing the follies and errors of human risk perception in behavioral economics, especially those risks that are rare, unfamiliar, or emotionally clouded. He wondered how informed consent could be preserved amidst these confounding factors and biases.
Commentary
A traditional ethical analysis of this case would point out that preservation of patient autonomy in the informed consent process is the paramount objective [1]. Mrs. Jones must be informed of all the pertinent risks, benefits, and alternatives to the treatment recommendation in order to make a fully informed and independent choice. A traditional approach might simply advise that, as an adult patient with decision-making capacity, Mrs. Jones has the right to refuse medical care even against the recommendations of the treating physician and family members [1].
Informed Consent and the Problem of Bias
However, informed consent and decision making are influenced by underlying cognitive processes, which may interefere with rational judgment. Under stressful and uncertain conditions, risk information is often misunderstood, and rational judgment can be obscured. Cognitive tendencies, limitations, and even errors can often influence judgment inappropriately, a problem known as “bias” [2, 3]. A growing body of literature suggests that cognitive biases and decision-making heuristics strongly influence decision making for both patients and physicians [2, 3]. Several types of bias may negatively impact a decision or outcome, particularly when the stakes are high and the risks uncertain.
Informational bias. To begin with, medical information can include implicit biases that often go unacknowledged but influence treatment decision making. Clinical studies may compare treatment modalities or select outcome measures or time points based on unjustified value assumptions [4]. For example, “unfavorable” neurological outcomes are traditionally designated in research studies by a Modified Rankin Score (mRS) greater than three, but whether or not individual patients define “unfavorable” in that way is unknown [5]. Studies that dichotomize results in this way may reflect research priorities rather than patient or clinician preferences. Moreover, academic medical centers are commonly the main sites for clinical research investigations and treatment recommendations, and this context may not reflect outcomes or available treatment options in nonacademic or community practice settings [6].
The effects of framing bias—including emphasizing only negative outcomes, focusing on the impact of disability, and selectively providing details about treatment options—have also been shown to influence patient decision making [7, 8]. Studies demonstrate that patient perception of prognostic information strongly influences decision making. Factors include the order in which negative information is presented, the time frame used for outcome assessment, the use of relative versus absolute risk reduction, the use of proportions instead of probabilities, and the inclusion of graphical materials [9, 10]. Poor statistical literacy among patients and physicians has also been reported as a barrier to the informed consent process [11].
The case scenario provides several examples of informational bias. Dr. Swan emphasized the risks of intervention (e.g., 15 percent chance of neurologic deficit, risk of death less than 1 percent) without ever reframing these statistics in more positive terms (e.g., 85 percent chance of no neurologic deficit, greater than 99 percent chance of survival after surgery). He also described the annual risk of aneurysmal rupture (i.e., 1 percent) without mentioning the much higher cumulative risk over Mrs. Jones’s lifetime. In the case, Mrs. Jones actually refuses surgery based upon the risk of neurological deficit associated with surgical intervention. Her perception of neurological risk might have changed had Dr. Swan presented the information differently.
Geographic bias. Geographic studies suggest that institutional factors and the local culture of clinical practice strongly influence physicians’ practice patterns. For example, the work of the Dartmouth Atlas group demonstrates wide geographic variation in procedural rates for hip and knee replacements, tonsillectomy, prostatectomy, cardiac surgery, and back surgery [12-15]. These variations appear to be associated with local physician supply and opinion and regional infrastructure and remain unexplained by patient-reported preferences for surgery or the prevalence of disease in a population [16].
This phenomenon has been observed in the treatment of cerebral aneurysms. Nonsurgical endovascular techniques for the treatment of cerebral aneurysms, developed over the past two decades, demonstrate similar efficacy and safety profiles as standard surgical approaches [17]. However, studies report wide institutional and regional variations in rates of endovascular procedures, with some regions reporting that they are used for 30 percent of all treated aneurysms and other regions reporting rates greater than 90 percent [18]. Much of the variation appears to be related to institutional factors such as physician expertise, equipment availability, and referral patterns between surgeon and nonsurgeon endovascular specialists [18].
In the case scenario, Dr. Swan consults the interventional neuroradiology team for nonsurgical treatment options but apparently does not mention to Mrs. Jones that the recommendation for surgical treatment involved other clinicians. He does not discuss the institutional factors and regional biases that structure his treatment decision and may even be unaware of the regional practice customs that influence his decision making. Dr. Swan’s own lack of endovascular expertise, the existing institutional referral patterns between neurosurgeons and neurointerventionalists, and regional practice patterns may all affect his treatment recommendation. In turn, Mrs. Jones’s limited knowledge of surgical and nonsurgical treatment techniques and outcomes influences her appraisal of the treatment recommendations. These factors may subtly influence the discussion of treatment options and risk perception in this case.
Individual bias. The presence of individual biases in medical decision making may seem particularly obvious and unremarkable. However, individual biases have been associated with very specific practice patterns, some related to life and death decisions. A study by Garland et al. [19] showed that decisions to limit life support were more strongly associated with the individual treating physician than with the patient’s comorbid conditions, diagnostic category, and source of ICU admission. Several studies demonstrate associations between a physician’s years in practice, attitudes toward medical care, and religious beliefs with a willingness to offer treatment or to withdraw care [20-22]. Clinicians also have professional biases. For example, prognostic accuracy is often influenced by patients’ conditions [23, 24], and the use of tests and procedures is associated with financial incentives [25]. Both patients and clinicians tend to focus on how life changes after a major health event without acknowledging aspects that remain unchanged, a cognitive bias known as the “focusing illusion” [23, 26, 27].
In the case scenario, little personal information is exchanged between Dr. Swan and Mrs. Jones that could help explain how indvidual biases might be influencing decision making and the informed consent process. What is Mrs. Jones’s understanding of neurologic deficits and the potential for rehabilitation when she says, “I wouldn’t want my children to see me like that”? The effect of a focusing illusion may be driving Mrs. Jones’s mostly negative perception of what her life would be like in the event of a neurological deficit after surgery. Moreover, Dr. Swan may be too focused on treating the aneurysm and preventing a rupture event, rather than appreciating Mrs. Jones’s concerns about neurological disability.
Relationship and the Limits of Autonomy
At its core, the problem raised by the case scenario has much to do with how we have come to model the informed consent process in medicine today. Under the current paradigm, the competent patient is considered an autonomous decision maker who must be provided with the relevant information and make an independent choice [28]. Whether or not the patient’s decision is in accord with the physician’s recommendation is largely irrelevant. The patient-physician relationship today is more contractual than fiduciary, more a matter of disclosure and fair exchange than trust.
However, our brief survey of the possible impact of bias on the informed consent process suggests that the expectation of completely autonomous decision making is somewhat unrealistic. From the moment a patient and physician meet, a torrent of cognitive, geographic, and personal factors surface that can confound risk perception and independent rational decision making. Mrs. Jones decides against treatment, focusing on the immediate risks of neurologic complications associated with surgical treatment. Dr. Swan emphasizes the potentially “catastrophic” risks of not treating the aneurysm. Doctor and patient interpret the same risk information from different perspectives and in different ways. Dr. Swan is committed to respecting Mrs. Jones’s wishes but remains concerned about her clinical benefit.
In this case scenario, the goal is to ensure that Mrs. Jones’s decision be informed and autonomous. But treatment decisions, particularly in high-risk situations, require more than simply exchanging information or eliciting preferences. A recent study by Sulmasy et al. demonstrated that patients with serious illnesses prefer a model of shared decision making, in which patient preferences and clinician recommendations carry equal weight in making a treatment decision [29]. Treatment decisions in high-risk situations require a dynamic relationship between doctor and patient in which patient preferences and clinician recommendations can interact equally to help shape a final treatment decision.
Mrs. Jones’s refusal of treatment is then not the endpoint of the case. The goal is to promote a shared decision between Dr. Swan and Mrs. Jones, in which Mrs. Jones continues to follow-up with Dr. Swan and discuss treatment options and goals of care. Dr. Swan should acknowledge the geographic and individual biases at work in his own treatment recommendation insofar as he is aware of them and provide risk information framed both positively and negatively. He might also provide graphical depictions of the medical risks in the form of bar or pie charts or icon-based pictographs to help Mrs. Jones understand such abstract information. If Mrs. Jones remains opposed to surgery, Dr. Swan should share his concern about that decision with her and ask her to return soon for more discussion. He cannot override her refusal and must avoid making Mrs. Jones feel that she is being coerced. If he believes that Mrs. Jones exposes herself to greater risk by doing nothing, he must hope that his routine followup and discussions with her—perhaps with new evidence of the aneurysm’s change—will cause her to reconsider her decision.
References
-
Beauchamp TL, Childress JF. Principles of Biomedical Ethics. 5th ed. New York, NY: Oxford University Press; 2001.
- Loewenstein G. Projection bias in medical decision making. Med Decis Making. 2005;25(1):96-105.
-
Kahneman D. Thinking, Fast and Slow. New York, NY: Farrar, Straus and Giroux; 2011.
-
Institute of Medicine (US) Forum on Drug Discovery, Development, and Translation. Challenges in clinical research. In: Institute of Medicine (US) Forum on Drug Discovery, Development, and Translation. Transforming Clinical Research in the United States: Challenges and Opportunities: Workshop Summary. Washington, DC: National Academies Press; 2010:14-28. http://www.ncbi.nlm.nih.gov/books/NBK50888/. Accessed October 26, 2014.
-
Puetz V, Campos CR, Eliasziw M, Hill MD, Demchuk AM; Calgary Stroke Program. Assessing the benefits of hemicraniectomy: what is a favourable outcome? Lancet Neurol. 2007;6(7):580.
- McGlynn EA, Asch SM, Adams J, et al. The quality of health care delivered to adults in the United States. N Engl J Med. 2003;348(26):2635-2645.
- Ainslie N, Beisecker AE. Changes in decisions by elderly persons based on treatment description. Arch Intern Med. 1994;154(19):2225-2233.
- Redelmeier DA, Rozin P, Kahneman D. Understanding patients’ decisions. Cognitive and emotional perspectives. JAMA. 1993;270(1):72-76.
- Epstein RM, Alper BS, Quill TE. Communicating evidence for participatory decision making. JAMA. 2004;291(19):2359-2366.
- Weeks JC, Cook EF, O’Day SJ, et al. Relationship between cancer patients’ predictions of prognosis and their treatment preferences. JAMA. 1998;279(21):1709-1714.
- Wegwarth O, Schwartz LM, Woloshin S, Gaissmaier W, Gigerenzer G. Do physicians understand cancer screening statistics? A national survey of primary care physicians in the United States. Ann Intern Med. 2012;156(5):340-349.
- Wennberg DE, Dickens JD Jr, Biener L, Fowler FJ Jr, Soule DN, Keller RB. Do physicians do what they say? The inclination to test and its association with coronary angiography rates. J Gen Intern Med. 1997;12(3):172-176.
- Wennberg J, Gittelsohn A. Small area variations in health care delivery. Science. 1973;182(4117):1102-1108.
- Wennberg JE, Blowers L, Parker R, Gittelsohn AM. Changes in tonsillectomy rates associated with feedback and review. Pediatrics. 1977;59(6):821-826.
- Wennberg JE, Gittelsohn A. Health care delivery in Maine I: patterns of use of common surgical procedures. J Maine Med Assoc. 1975;66(5):123-130, 149.
-
Wennberg JE. Time to tackle unwarranted variations in practice. BMJ.2011;342:d1513. doi:10.1136/bmj.d1513.
- Spetzler RF, McDougall CG, Albuquerque FC, et al. The Barrow Ruptured Aneurysm Trial: 3-year results. J Neurosurg. 2013;119(1):146-157.
-
Bekelis K, Goodney PR, Dzebisashvili N, Goodman DC, Bronner KK. Variation in the care of surgical conditions: cerebral aneurysm. Dartmouth Atlas of Health Care; 2014. http://www.dartmouthatlas.org/downloads/reports/Cerebral_aneurysm_report_09_30_14.pdf. Accessed October 26, 2014.
- Garland A, Connors AF. Physicians’ influence over decisions to forego life support. J Palliat Med. 2007;10(6):1298-1305.
- Christakis NA, Asch DA. Biases in how physicians choose to withdraw life support. Lancet. 1993;342(8872):642-646.
- Christakis NA, Asch DA. Physician characteristics associated with decisions to withdraw life support. Am J Public Health. 1995;85(3):367-372.
- Christakis NA, Lamont EB. Extent and determinants of error in doctors’ prognoses in terminally ill patients: prospective cohort study. BMJ. 2000;320(7233):469-472.
- Creutzfeldt CJ, Holloway RG. Treatment decisions after severe stroke: uncertainty and biases. Stroke. 2012;43(12):3405-3408.
- Finley Caulfield A, Gabler L, Lansberg MG, et al. Outcome prediction in mechanically ventilated neurologic patients by junior neurointensivists. Neurology. 2010;74(14):1096-1101.
- Emanuel EJ, Fuchs VR. The perfect storm of overutilization. JAMA. 2008;299(23):2789-2791.
- Kahneman D, Krueger AB, Schkade D, Schwarz N, Stone AA. Would you be happier if you were richer? A focusing illusion. Science. 2006;312(5782):1908-1910.
- Swindell JS, McGuire AL, Halpern SD. Shaping patients’ decisions. Chest. 2011;139(2):424-429.
- Sulmasy DP, Snyder L. Substituted interests and best judgments: an integrated model of surrogate decision making. JAMA. 2010;304(17):1946-1947.
- Sulmasy DP, Hughes MT, Thompson RE, et al. How would terminally ill patients have others make decisions for them in the event of decisional incapacity? A longitudinal study. J Am Geriatr Soc. 2007;55(12):1981-1988.