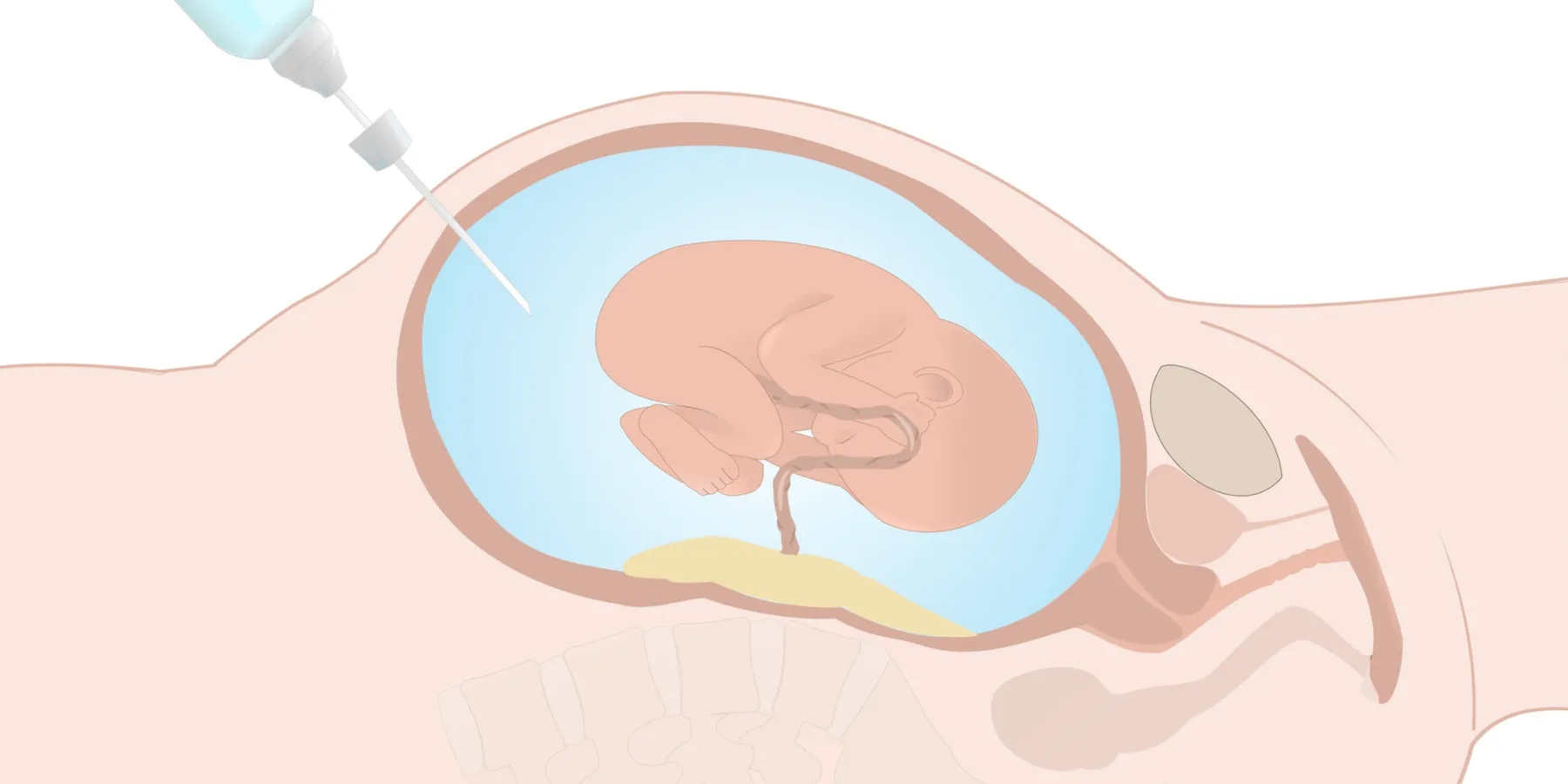
Amniocentesis is an invasive procedure that requires removing a sample of amniotic fluid to obtain fetal cells for chromosome analysis. Generally not performed earlier than 15 weeks gestation, the procedure is done under ultrasound guidance. Although some pain is associated with amniocentesis, it is generally well tolerated without the need for anesthesia. Five to 10 percent of pregnant women choose to have the test [1, 2]. Maternal age over 35 is the most common current indication for amniocentesis [3].
Amniocentesis was developed in the 1960s, and until 2007 it was offered almost exclusively to women with an identified risk for carrying a fetus with a genetic disorder that could be detected by traditional karyotyping. This included women over 35, those with a previously affected child, those with a family history of a genetic disorder, and those with a serum screening positive for Down syndrome or trisomy 18. Amniocentesis was also offered to women who showed fetal structural irregularities on ultrasound that could be markers for chromosomal abnormalities.
Counseling a patient about the risks and limitations of the test is essential, both before and after the procedure. Some women—those who, for example, would not choose to terminate their pregnancy under any circumstances—may feel that an invasive test is unnecessary, particularly if they see no advantage to knowing about a genetic disease before the baby's birth. Other women may want to proceed with an amniocentesis to prepare for the care of an affected child, even if they would not choose to terminate the pregnancy.
The indications for offering amniocentesis were revised by the American College of Obstetricians and Gynecologists (ACOG) in a 2007 practice bulletin that included a new recommendation for Down syndrome screening [4]. The new standard of care no longer uses maternal age as the basis for deciding whether to offer genetic screening or other invasive tests like amniocentesis. Rather, ACOG's goal is to offer screening tests with high detection and low false positive rates to all women [5]. The 2007 bulletin advises physicians to counsel patients of all ages about the risks and benefits of genetic testing and to let the patient decide whether the benefits of obtaining the test results are worth the risks. In short, the goal is to make all tests for fetal chromosome abnormalities more accessible to all women. On a practical level, physicians should inform their patients that not all health insurance companies cover procedures like amniocentesis for women under 35 who have no risk factors.
Risks Associated with Amniocentesis
Amniocentesis was not offered to all women in the past because the increased risk of pregnancy loss after the invasive procedure did not seem balanced by the potential for benefit among women at low risk for having a fetus with an abnormal karyotype. As recently as 10 years ago, the risk for pregnancy loss after amniocentesis was thought to be approximately the same as the baseline probability that a 35-year-old woman would have a child with Down syndrome—about 0.5 percent or 1 in 200 [6].
The risk of injury to the mother or fetus during amniocentesis is extremely low. The primary concern that should be conveyed to patients is the risk of miscarriage after the procedure, but that risk is difficult to calculate because studies with adequate controls are lacking and because risk of miscarriage is already higher for most of the women who choose to undergo amniocentesis.
A 2007 systematic review of data compiled from 29 studies of amniocentesis found that the risk of miscarriage within 14 days of the procedure was 0.6 percent, pregnancy loss before 24 weeks (the age when a fetus is considered to be viable) was 0.9 percent, and the total pregnancy loss at any point following amniocentesis was 1.9 percent [7]. Even with these general percentages, the authors of the review article caution that the "lack of adequate controls tends to underestimate the true added risk of prenatal invasive procedures" [8]. A Cochrane review of 14 randomized studies that compared the safety of amniocentesis and chorionic villus sampling (another form of prenatal diagnosis) found that amniocentesis increased the already-known 2 percent risk of pregnancy loss by 1 percent [9]. The controversy continues with a 2008 report from a single institution documenting more than 50,000 cases (with controls) over 16 years and indicating an amniocentesis-related pregnancy loss of 0.13 percent [10].
Benefits of Amniocentesis
The benefits of amniocentesis are numerous. The results may reassure an anxious couple that the fetus has normal chromosomes. Conversely, it may confirm a suspicion raised by ultrasound or serum screening and help the couple decide whether to terminate the pregnancy or continue and prepare for the unique needs of the child. Genetic testing performed on the cells extracted during amniocentesis does not eliminate the possibility of significant or lethal structural anomalies, but the latter are easily found on a detailed anatomy ultrasound.
Amniocentesis has an essential place in prenatal genetic diagnosis, although it is not without risk. Obstetricians should offer screening to all patients and have a frank discussion about how the knowledge will help the couple and about the small, but real, risks involved in acquiring that knowledge.
References
- Nicolaides KH, Chervenak FA, McCullough LB, Avgidou K, Papageorghiou A. Evidence-based obstetric ethics and informed decision-making by pregnant women about invasive diagnosis after first-trimester assessment of risk for trisomy 21. Am J Obstet Gynecol. 2005;193(2):322-326.
- Steele MW, Berg WR Jr. Chromosome analysis of human amniotic fluid cells. Lancet. 1966;1(7434):383-385.
-
Papp C, Papp Z. Chorionic villus sampling and amniocentesis: what are the risks in current practice? Curr Opin Obstet Gynecol. 2003;15(2):159-165.
-
ACOG Committee on Practice Bulletins. ACOG Practice Bulletin No. 77: Screening for fetal chromosomal abnormalities. Obstet Gynecol.2007;109(1):217-228.
-
ACOG, 222.
-
Centers for Disease Control and Prevention. Chorionic villus sampling and amniocentesis: recommendations for prenatal counseling. MMWR Recomm Rep. 1995;44:1-4.
- Mujezinovic F, Alfirevic Z. Procedure-related complications of amniocentesis and chorionic villus sampling: a systematic review. Obstet Gynecol. 2007;110(3):687-694.
-
Mujezinovic, 687.
-
Alfirevic Z, Sundberg K, Brigham S. Amniocentesis and chorionic villus sampling for prenatal diagnosis. Cochrane Database Syst Rev. 2003;3:CD003252.
- Odibo AO, Gray DL, Dicke JM, Stamilio DM, Macones GA, Crane JP. Revisiting the fetal loss rate after second-trimester genetic amniocentesis: a single center's 16-year experience. Obstet Gynecol. 2008;111(3):589-595.