Abstract
Background: Financial incentives have been shown to improve antiretroviral (ARV) adherence for people living with HIV, but scholars have argued that this commodifies treatment and have debated the ethics of doing so. This article summarizes research on ethical processes and factors involved in an intervention that successfully improved ARV adherence among socially vulnerable people living with HIV.
Methods: Thirty qualitative interviews were conducted with intervention participants and field notes documenting organizational processes were analyzed. The protocol utilized a preexisting framework to assess the ethics of using financial incentives to motivate adherence.
Results: Financial incentives supported an ethical service provision framework by (1) establishing and strengthening client agency, (2) revising organizational protocols to prioritize adherence, and (3) promoting resource redistribution.
Conclusions: Financial incentives, when embedded in wrap-around services, innovative client-centered organizational processes, and a justice orientation, constitute an ethical intervention requiring ethical investigation.
Introduction
Financial incentives have been shown to be an effective intervention to boost antiretroviral (ARV) adherence among people living with HIV (PWH).1,2,3,4,5 Examining the effects of combining intensive case management services to maintain adherence with financial incentives ($100 gift voucher for viral load < 50c/ml at quarterly assays), Ghose et al found that the Undetectables Intervention (UI) significantly improved ARV adherence and maintained it over a 4-year period in a sample of socially vulnerable PWH with a high prevalence of homelessness, substance use, and mental illness.6
Despite the proven effectiveness of paying participants to maintain medication adherence and achieve wellness goals, the ethics of financial incentives have been debated. Scholars have expressed concern that financial incentives coerce participation, exacerbate inequities, subject life and health to valuation processes, undermine therapeutic relationships, and prioritize financial over health outcomes.7,8,9,10,11 Drawing on a framework put forth by Christensen12 that describes an ethical orientation to community mental health service provision, Claassen argues that the provision of incentives needs to take into account: (1) complications in the process of informed consent, (2) the possibility of incentives being paternalistic and coercive, (3) issues of resource allocation that address organizational resource capacity and what constitutes optimal incentives, (4) organizational relationships shaped by this incentive, (5) beneficence, and (6) nonmaleficence.13
Extending the previous study by Ghose et al,6 this qualitative study examines the ethical dimensions of the financial incentives used in the UI. We apply Claassen’s adaptation of Christiansen’s framework to examine and interpret our results.
Methods
We conducted 30 semistructured qualitative interviews with UI participants recruited through snowball sampling to examine the ways financial incentives influence adherence-related behaviors. Of the final sample of 30 interviewees, 77% were African American, 13% were Latinx, and 10% were Asian American or White and 43% were men and 57% were women. We used NVivo (N6) to analyze the data. Interviews were discontinued when conceptual saturation was reached, whereby new concepts ceased to emerge from coded data. We also used field notes from service provision team meetings and trainings to support concepts. Drawing on a grounded theory approach using sensitizing concepts,14 thematic analyses were employed to identify concepts and themes.
Results
Our results indicate that financial incentives support an ethical service provision framework by (1) establishing and strengthening client agency, (2) revising organizational protocols to prioritize adherence, and (3) promoting principles of redistributional justice. We found that the principle of beneficence infuses each of the 3 ethical dimensions (see Figure).
Figure. An Ethical Framework for Considering the Delivery of Financial Incentives
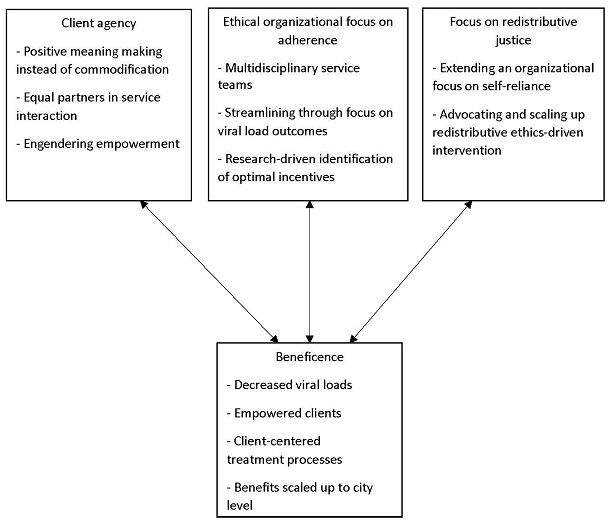
Developing client agency. When examining the interview data for evidence of Christensen’s first 2 categories, informed consent and paternalism, we found that these concepts merged into a consolidated theme of developing and strengthening client agency. Participants reported that, with financial incentives, they felt less stigmatized or pressured to take medicine. One participant compared medication decisions to making decisions about work, stating: “Now I take it, that’s great; if I don’t, there’s no shame in it, I just don’t get it (the voucher). It’s like taking a day off from work.” Another reiterated this point by opining, “It makes me feel like I’m an adult. Not someone who is forgetting and has to be told about it constantly, but someone who is being paid to take the meds. That’s cool and makes me OK if I miss it here or there.”
This nonstigmatizing, nonpaternalistic orientation to missed doses allows participants to overcome shame and stay adherent. Being able to maintain adherence over the long term despite some missed doses is significant, given the evidence that “drug holidays” (whereby some doses are missed while otherwise being adherent) do not contribute to increased viral loads.15,16
Ghose et al found that, far from commodifying the act of taking medicine, incentives infused it with meaning. In particular, participants felt that the incentives acknowledged their participation on the front lines of the campaign to bring the HIV epidemic to an end.6 We found that this meaning-making process significantly empowered participants in the clinician-client relationship by underlining the role of the participant as an equal partner in the health service interaction. As one participant put it: “I’m not paying you, I’m being paid. That means I have something to give. I’m not just being helped, I am also helping.” The development and strengthening of participant agency mitigates the possibility that informed consent was undermined or that clients were treated paternalistically. In fact, the incentives facilitated a decision-making process that incorporated participants’ essential role in the fight against the epidemic. Rather than becoming objects of an intervention, participants championed their subjectivity in the act of being paid. In addition, the strengthening of client autonomy was infused by beneficence, demonstrated tangibly through viral load suppression and subjectively through the way participants found their voice as partners in the interaction with clinicians.
Far from commodifying the act of taking medicine, incentives infused it with meaning.
Ethical organizational protocols to prioritize adherence. Christiansen’s and Claassen’s concepts of allocation of organizational resources and of organizational relationships or boundaries12,13 merged into a theme we classified as emergence of ethical organizational practices that focused on adherence.
The relatively poor ARV adherence rates in the socially precarious community served by the organization where participants were receiving their ARV treatment led to a revamping of organizational priorities. The organization sought to improve adherence rates to bring them on par with those for PWH in the larger community, despite the demonstrated barriers of substance use, homelessness, and mental illness that confronted its clients. Previously, the organization had focused on housing homeless people living with HIV. While housing addressed one of the most significant barriers to adherence, the implicit assumption that high adherence would be the eventual outcome was not borne out across all populations of clients. Accordingly, the organization refocused on ARV adherence, ushering in tangible viral load-related goals and innovative methods to incentivize adherence. We argue that this focus on adherence for the population that was at highest risk of nonadherence brought with it an ethical clarity that enabled streamlining of organizational practices. The organization felt that higher adherence would improve individual and public health outcomes and reduce avoidable health care costs to such an extent that paying for adherence would ultimately be cost-effective, significantly beneficial for participants who could thereby achieve and sustain viral suppression, and a meaningful strategy to address persistent HIV health inequities.
This was the logic used in securing funding from a private foundation to conduct a demonstration program to establish the feasibility of an ethical and effective intervention. An incentive of $100 per quarter for suppressed viral load count emerged as the optimal amount based on literature indicating that this incentive would be meaningful, noncoercive, and cost-effective.17,18,19 Participants routinely indicated that the incentives were meaningful in maintaining adherence. One participant noted: “It helps that me taking my meds also helps to pay for groceries.” Another noted that the incentives were also a way for the organization itself to resist inadvertently contributing to the illegal market in pills: “There’s always been these pharmacies [that would buy the pills]. This [payment as an incentive] just means that I can make money above the table, and not illegally. This helps.”
In addition to allocating organizational resources, the organization implemented several innovative strategies to support the incentives-based intervention. A social marketing campaign framed ARV adherence as a heroic accomplishment supporting individual health. Ghose et al noted that the campaign successfully motivated participants to enroll in the intervention.6 Service provision teams were formed comprising doctors, nurses, social workers, and case managers. Regular case conferences of the service provision team and the client were initiated to identify barriers to adherence and craft client-centered adherence supports. Monthly viral load suppression meetings were held in which service provision teams discussed progress and challenges, shared strategies of success, and engaged in multidisciplinary training. Often, the multidisciplinary audience would provide written and verbal feedback during these training sessions on how the intervention was forcing them to learn innovative techniques and pushing them to engage with other disciplinary perspectives to provide more integrated care. The organization also collaborated with academic partners to monitor outcomes and train service provision teams in evidence-based practices such as motivational interviewing (MI) and cognitive behavioral therapy (CBT).
On several occasions, case managers in the meetings would tackle the specter of unintended harm, especially for those who could not remain adherent. Christiansen flags this ethical concept of maleficence as one to be addressed explicitly in community health care provision.12 Training sessions addressed these concerns by encouraging health care professionals to work with clients following a stages-of-change (SOC) model, whereby success was defined by moving to the next stage rather than by moving immediately to the ultimate goal. Financial incentives were therefore a final reward within the SOC model. In other words, failure was removed from the equation as clients could take their own time to get to the final stage, given where they started. This approach undermined distinguishing those whose viral loads were undetectable from those whose viral loads were not. In one of the training sessions, the medical director for the organization, who was supervising the overall intervention, commented that it was refreshing to learn about evidence-based social work techniques of engaging with clients through MI, CBT, and SOC techniques. These innovative processes had always been aspirational goals. The incentive initiative, with its measurable objective of viral load suppression and its financial investment in client success, appeared to galvanize the organization to concretize what had previously been merely aspirational goals for organizational process.
The innovative practices wrapped around financial incentives also inscribed beneficence into organizational practices, resulting in a systematized interdisciplinary culture that lives on in the organization. For instance, these practices have now been replicated at the organization in an intervention for promoting treatment engagement and adherence to medication for people living with hepatitis C. Initial results indicate that the initiative has resulted in significantly increased rates of enrollment in care, testing, adherence, and cure.
Focus on redistributive justice. Organizational staff advocated for financial redistributive justice more broadly following the launch of the incentive program. City officials recognized that redistributive justice—in transferring money directly to participants with the goal of addressing social barriers to adherence and resulting health inequities—was informed by egalitarianism and a nonpaternalistic orientation. Based on the initial success of the intervention, this message of a client-centered incentives approach hit its mark when the city’s department of health funded a scale-up of the program to include other agencies seeking to improve HIV treatment effectiveness among vulnerable PWH.20
It is important to note here that though redistributive justice through the mechanism of financial incentives was a new mode of patient advocacy, the organization’s activities were already rooted in principles of client self-reliance and economic justice. Formed as a membership organization of clients, staff, and volunteers, the organization has a strong commitment to advocacy with and on behalf of low-income PWH. It also has a well-established practice of hiring clients who have graduated from its job-training programs for staff positions, supporting their ongoing education, and promoting them through the ranks. From its inception, the organization has employed social enterprise, including upscale thrift shops, to generate funding for its operations and provide employment opportunities for clients. The use of financial incentives, therefore, was both an outgrowth of applying the organization’s principles of client self-reliance and economic justice and an accelerant for seeking broader redistributive justice.
A citywide scale-up of financial incentives for ARV adherence, based on the same innovative and collaborative practices that made the intervention effective within the organization, helped to promote justice by spreading benefits to similar populations beyond the organization.
Conclusion
Our results indicate that financial incentives, when embedded in wrap-around services, innovative client-centered organizational processes, and a justice orientation, constitute an ethical intervention. Beneficence was infused in every element of the framework, as discussed above. While we found evidence for some of Christiansen’s and Claassen’s categories of ethical service provision, new themes emerged in our data. Specifically, we found that by emphasizing factors of ethical concern in financial incentives, Claassen’s framework, with its emphasis on paternalism and coercion, undertheorizes the possibility of positive ethical outcomes in providing financial incentives. In particular, we found that incentives trigger innovative processes on the personal, organizational, and citywide levels, all of which strengthen ethical treatment. Our results support a conceptual ethical framework for assessing incentives-based interventions and other interventions in the field of HIV care.
References
-
El-Sadr WM, Donnell D, Beauchamp G, et al; HPTN 065 Study Team. Financial incentives for linkage to care and viral suppression among HIV-positive patients: a randomized clinical trial (HPTN 065). JAMA Intern Med. 2017;177(8):1083-1092.
- Adamson B, El-Sadr W, Dimitrov D, et al. The cost-effectiveness of financial incentives for viral suppression: HPTN 065 study. Value Health. 2019;22(2):194-202.
-
Greene E, Pack A, Stanton J, et al. “It makes you feel like someone cares”: acceptability of a financial incentive intervention for HIV viral suppression in the HPTN 065 (TLC-Plus) study. PLoS One. 2017;12(2):e0170686.
- Brantley AD, Burgess S, Bickham J, Wendell D, Gruber D. Using financial incentives to improve rates of viral suppression and engagement in care of patients receiving HIV care at 3 health clinics in Louisiana: the Health Models Program, 2013-2016. Public Health Rep. 2018;133(2)(suppl):75S-86S.
- Silverman K, Holtyn AF, Rodewald AM, et al. Incentives for viral suppression in people living with HIV: a randomized clinical trial. AIDS Behav. 2019;23(9):2337-2346.
- Ghose T, Shubert V, Poitevien V, Choudhuri S, Gross R. Effectiveness of a viral load suppression intervention for highly vulnerable people living with HIV. AIDS Behav. 2019;23(9):2443-2452.
- Priebe S, Sinclair J, Burton A, et al. Acceptability of offering financial incentives to achieve medication adherence in patients with severe mental illness: a focus group study. J Med Ethics. 2010;36(8):463-468.
-
Noordraven EL, Schermer MHN, Blanken P, Mulder CL, Wierdsma AI. Ethical acceptability of offering financial incentives for taking antipsychotic depot medication: patients’ and clinicians’ perspectives after a 12-month randomized controlled trial. BMC Psychiatry. 2017;17(1):313.
- Madison KM, Volpp KG, Halpern SD. The law, policy, and ethics of employers’ use of financial incentives to improve health. J Law Med Ethics. 2011;39(3):450-468.
- Lunze K, Paasche-Orlow MK. Financial incentives for healthy behavior: ethical safeguards for behavioral economics. Am J Prev Med. 2013;44(6):659-665.
- Won T, Blumenthal-Barby J, Chacko M. Paid protection? Ethics of incentivised long-acting reversible contraception in adolescents with alcohol and other drug use. J Med Ethics. 2017;43(3):182-187.
- Christensen RC. Ethical issues in community mental health: cases and conflicts. Community Ment Health J. 1997;33(1):5-11.
- Claassen D. Financial incentives for antipsychotic depot medication: ethical issues. J Med Ethic. 2007;33(4):189-193.
- Bowen GA. Grounded theory and sensitizing concepts. Int J Qual Methods. 2006;5(3):12-23.
- Kingsley L, Jacobson LP. Determinants of heterogeneous adherence to HIV-antiretroviral therapies in the Multicenter AIDS Cohort Study. J Acquir Immune Defic Syndr. 2001;26(1):82-92.
-
Parienti JJ, Ragland K, Lucht F, et al; ESPOIR and REACH study groups. Average adherence to boosted protease inhibitor therapy, rather than the pattern of missed doses, as a predictor of HIV RNA replication. Clin Infect Dis. 2010;50(8):1192-1197.
- Farber S, Tate J, Frank C, et al. A study of financial incentives to reduce plasma HIV RNA among patients in care. AIDS Behav. 2013;17(7):2293-2300.
- Goldie SJ, Paltiel AD, Weinstein MC, et al. Projecting the cost-effectiveness of adherence interventions in persons with human immunodeficiency virus infection. Am J Med. 2003;115(8):632-641.
-
Schackman BR, Finkelstein R, Neukermans CP, Lewis L, Eldred L; Center for Adherence Support and Evaluation (Case) Team. The cost of HIV medication adherence support interventions: results of a cross-site evaluation. AIDS Care. 2005;17(8):927-937.
- Gambone GF, Feldman MB, Thomas-Ferraioli AY, Shubert V, Ghose T. Integrating financial incentives for viral load suppression into HIV care coordination programs: considerations for development and implementation. J Public Health Manag Pract. 2020;26(5):471-480.