Case
Dr. Jorgensen is a family practice doctor with a steady, yet varied, patient population. Practicing medicine in a small suburban community, he sees whole families ranging from newborn babies to adults well into their 80s. He prides himself on his practice of preventive medicine in particular, and, because he has such a longstanding rapport with his patients, they adhere to his counseling on healthy living and follow up regularly for annual physicals and appropriate screening tests.
Among other things, Dr. Jorgensen is particularly diligent in his screening for, and treatment of, diabetes mellitus; he tests all of his patients over age 45 for diabetes and refers all of his diagnosed diabetic patients for annual ophthalmologic exams.
Recently, Dr. Jorgensen began mentoring a new family practice physician, Dr. Sandkey. Dr. Sandkey completed her residency in family practice at a large, inner-city academic hospital, where she attended a number of lectures on cost-effective treatment. Like Dr. Jorgensen, Dr. Sandkey has a special interest in preventive medicine and is well-versed in the current recommendations regarding diabetes screening and treatment.
Because Dr. Sandkey is looking to model her practice after Dr. Jorgensen’s, she has been going through old patient charts to see how Dr. Jorgensen tracks his patients’ health maintenance. One day she approaches Dr. Jorgensen to discuss his screening practices: “Dr. Jorgensen, I have to ask you about how you choose your medical interventions and screenings. I read a study that indicated screening everyone age 45 and older for diabetes had minimal benefit but cost more than $500 per person on average. Why have you decided to screen this entire group of patients?”
Commentary
“The human condition is such that…there are many possible courses of actions and forms of life worth living, and therefore to choose between them is part of being rational or capable of moral judgment; [we] cannot avoid choice for one central reason...namely that ends collide; that one cannot have everything....The very concept of an ideal life...is not merely utopian, but incoherent.”
Isaiah Berlin [1]
Dr. Jorgenson is imbued with a sense of obligation to his patients that transcends matters of cost. He wants to serve them well within a society that can seem frivolous in its willingness to indulge the “haves” and penurious when it comes to the health and well-being of the “have nots.”
Let’s say that Dr. Jorgensen graduated in the 1960s from an institution known for its difficulty and its superb reputation for training clinicians. In Dr. Jorgensen’s day, students spent the first 2 years deeply engaged in the fundamentals of biochemistry, physiology, and other building blocks needed to undergird the practice of clinical medicine, which were the emphasis of the third and fourth years.
If the word “ethics” was spoken in a class or clinical setting, Dr. Jorgensen does not remember. But to him, ethical practice means providing the same care for rich and poor, powerful, and disenfranchised alike. In the current American health context, in which diabetes is a real risk for many, including or especially the poor, Dr. Jorgenson understands equal care to mean putting a particular emphasis on diabetes screening, as recommended by the American Diabetic Association [2].
Fresh from her much more recent education and residency in family medicine at Dr. Jorgenson’s alma mater, Dr. Sandkey’s training very likely included an emphasis on cost effectiveness—overtly, in the form of lectures and seminars, and more subtly, in the culture of her educational institutions. The difference in their perspectives may be due more to the evolving priorities of medical education than to a lack of concern for patients on her part. But Dr. Sandkey could put Dr. Jorgenson off by broaching this topic as though cost effectiveness is the primary concern. If anything, even if she came with the highest levels of recommendation for her engagement, understanding, and skills, it could make Dr. Jorgenson question his judgment in choosing her as a mentee. “What a mistake,” he might think. “The product of an enlightened education comes out worshiping the almighty dollar rather than caring about the essential needs of patients. I’m ashamed.”
Dr. Sandkey might foster a more productive conversation if she takes a respectful, evidence-based approach and reassures Dr. Jorgenson of her commitment to the patients’ interests. Supposing Dr. Sandkey said the following: “Preventive medicine is one of my highest priorities. What I’m suggesting is only that there may be an alternative way to provide optimal service at the lowest cost and danger. The ‘population strategy’ you suggest for screening, in which we screen everyone, is best when there is a diagnosable and treatable problem with few signs or symptoms spread though the community which we have a low-cost, low-pain method of identifying. As Caroline Wright has written, ‘organized population screening programs [must be] designed to ensure that the benefits of screening outweigh the harms’ [3]. Screening babies for hearing loss and infants for lead poisoning meets those standards [4]. I’m not sure that a population strategy serves our older patients who might have diabetes.”
Dr. Sandkey is in favor of at “at-risk” strategy, one in which clinicians identify the presence of “biologic or environmental factors that predispose to disease…[and] easily recognizable early warning signs that [it] is impending,” and screen only the patients who are subject to those factors [5]. She suggests, “An ‘at-risk’ strategy would work better, especially if combined with guidance given to everyone—a ‘public health’ strategy” [6]. This last approach, often used in childhood, is not to screen at all because the risks of screening are too high. Instead, everyone receives recommendations for healthy living [7].
Dr. Sandkey might support her argument by respectfully mentioning that her ideas are in agreement with evidence-based guidelines made to further the interests of patients. The United States Preventive Services Task Force, for example, recommends an “at-risk” group approach for diabetes screening [8]. Though the ADA takes a population approach [1], it adds “particularly those with a body mass index of 25 kilograms per meter squared or greater.” Emphasis is added to show that the ADA leaves discretion for use of an “at-risk” approach to the clinician. As its data show, there was moderate evidence of effectiveness only for screening adults with hypertension.
Dr. Sandkey could say, “Both the American Academy of Family Physicians [9] and the Canadian Task Force on Preventive Health Care [10] found insufficient evidence to recommend screening adults who are at low risk for coronary vascular disease. This seems like a good way of removing patients who are very unlikely to have a positive result from the screening pool.”
Dr. Sandkey might go on to argue that screening should be limited to conditions that meet the following five criteria [11]:
- It is an important public health concern;
- there is an asymptomatic period;
- an effective screening test exists;
- there is a treatment for the disorder; and
- treating the asymptomatic stage is proven to provide long-term benefit.
Screening an undifferentiated population leads to an increase in the number of patients with false positive test results and a decrease in the positive predictive value of your testing [12]. According to Bayes’ Theorem, the predictive value (PV) of a test is proportional to the prevalence of the problem in the population surveyed. Thus, the goal of a screening process is to create the smallest possible pool of patients containing all or almost all affected individuals (the true positives, or TPs). An ideal—and ideally cost-effective—screening test has maximal sensitivity with least loss of specificity: the number of false positives (FPs) is kept at a minimum and the PV (TP/ [TP + FP]) is at its maximum. An appreciation of Bayes’ Theorem, often difficult for the clinician, is critical to the use of evidence-based medicine [13].
“Our goal,” she could declare, “is to create the smallest pool containing all or almost all who are affected, leaving out those we are sure are not.” And then she could show him this figure:
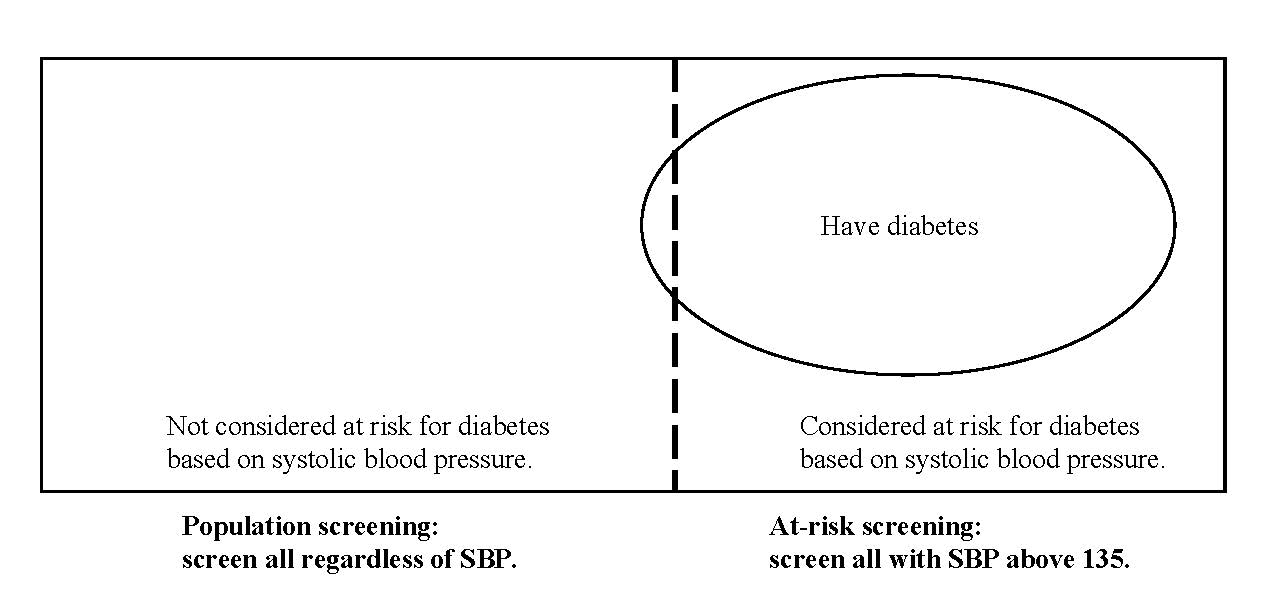
Fig. 1. Screening everyone (the entire rectangle) is a “population” strategy. Screening to the right of the dotted line would be taking an “at-risk” strategy.
“So,” she would continue, “an ‘at-risk’ approach is most efficacious and has the lowest human, as well as economic, cost. Of course, I do propose implementing a public health strategy: Though different treatments would be provided for those patients who tested positive and those who did not; preventive guidance is appropriate for everyone.”
By making clear her shared commitment to benefiting patients, Dr. Sandkey can show Dr. Jorgenson that she is interested in preventive medicine and well-versed in the recommendations for screening strategies, and he may be more willing to consider an alternative plan. They will do regular risk assessment interviews for all patients using a behavior modification approach that encourages healthful diet and habits. They will focus on obesity, smoking, and preventing hypertension. When, however, patients show identifiable risk for diabetes, they will perform formal testing.
Now they will be satisfied. Why? Because they were able to listen to each other’s concerns, communicate effectively, and find a common path that allows them both to maintain their ethical standards, and, finally, they can feel confident that they are doing right by their patients.
References
-
Berlin I. Four Essays on Liberty. Oxford: Oxford University Press; 1990.
-
American Diabetes Association. Standards of medical care in diabetes—2007. Diabetes Care. 2007;30 Suppl 1:S4-S41.
-
Wright C. Risks and benefits of population screening. Genome Unzipped. http://www.genomesunzipped.org/2010/07/risks-and-benefits-of-population-screening.php. Accessed March 11, 2011.
- Karp R, Abramson J, Clark-Golden M, et al. Should we screen for lead poisoning after 36 months of age? Experience in the inner-city. Ambulatory Pediatr. 2001;1(5):256-258.
- Jelliffe DB, Jelliffe EFP. The “at-risk” concept and young child nutrition programmes (practices and principles). J Trop Pediatr Environ Child Health. 1972;18(3):199-201.
- Wadowski S, Karp R, Senft C, Murray-Bachmann R. Family history of coronary artery disease and cholesterol screening in a disadvantaged population. Pediatrics. 1994;93(1):109-113.
- Newman TB, Berowner WS, Hulley SB. The case against childhood cholesterol screening. JAMA. 1990;264(23):3039-3043.
-
US Preventive Services Task Force. Screening for type 2 diabetes mellitus in adults; 2008. http://www.uspreventiveservicestaskforce.org/uspstf/uspsdiab.htm. Accessed March 11, 2011.
-
American Academy of Family Physicians. Summary of recommendations for clinical preventive services; 2007. http://www.aafp.org/online/etc/medialib/aafp_org/documents/clinical/clin_recs/cps.Par.0001.File.tmp/August2006CPS.pdf. Accessed March 11, 2011.
-
Feig DS, Palda VA, Lipscombe L; Canadian Task Force on Preventive Health Care. Screening for type 2 diabetes mellitus to prevent vascular complications: updated recommendations from the Canadian Task Force on Preventive Health Care. CMAJ. 2005;172(2):177-180.
-
Wilson JM, Junger G. Principles and Practice of Screening for Disease. Geneva: World Health Organization; 1968. http://whqlibdoc.who.int/php/WHO_PHP_34.pdf. Accessed March 11, 2011.
- Karp RJ. The ‘at-risk’ concept as applied to the identification of malnourished hospitalized patients: how a two-step process improves clinical acumen. Nutr Clin Pract. 1998;3(4):150-153.
- Christakis DA. Predictably unhelpful: why clinicians do not use prediction rules. Arch Pediatr Adolesc Med. 2011;165(1):90-91.