Abstract
Counseling patients regarding the benefits, harms, and dilemmas of genetic testing is one of the greatest ethical challenges facing reproductive medicine today. With or without test results, clinicians grapple with how to communicate potential genetic risks as patients weigh their reproductive options. Here, we consider a case of a woman with a strong family history of early-onset Alzheimer’s disease (EOAD). She is early in her pregnancy and unsure about learning her own genetic status. We address the ethical ramifications of each of her options, which include genetic testing, genetic counseling, and termination versus continuation of the pregnancy. Our analysis foregrounds clinicians’ role in helping to ensure autonomous decision making as the patient reflects on these clinical options in light of her goals and values.
Case
During his third-year OB-GYN clerkship in medical school, Samuel is working with Dr. Bowers seeing patients both in the hospital (on the labor and delivery service) and in the outpatient clinic for routine prenatal visits. For the outpatient visits, he sees patients who present for initial appointments to confirm pregnancy and for appointments just prior to delivery.
About halfway through his clerkship, Samuel and Dr. Bowers see Mrs. Castle and her husband for an initial visit to confirm a pregnancy. Mrs. Castle is a healthy 41-year-old woman with a strong family history for early-onset Alzheimer’s dementia. Her father was diagnosed with Alzheimer’s dementia at age 45 and died about five years later. Mrs. Castle’s older sister, who is in her late 40s, has also been diagnosed with early-onset Alzheimer’s dementia and is currently living in long-term care due to complications of the disease.
Mrs. Castle and her husband had tried to conceive for more than a year without success. They had met with a specialist and briefly considered assisted reproductive technology when they put their plans on hold due to Mrs. Castle’s sister’s illness. The couple thought carefully about what a pregnancy would mean for Mrs. Castle and her family; they chose not to pursue genetic counseling related to Mrs. Castle’s family history of early-onset Alzheimer’s dementia. Recently, Mrs. Castle has been more concerned about her own worsening short-term memory. Her husband has also noticed that she seems to have become more irritable and anxious over the last couple of years. They question whether they should continue with the pregnancy.
Dr. Bowers wondered how best to provide support for the patient not only in prenatal care but also in addressing the couple’s concerns about parenting in the setting of a strong family history of early-onset Alzheimer’s dementia.
Commentary
Dr. Bowers, Mrs. Castle, and her family all face a daunting set of decisions, as Mrs. Castle is early in her pregnancy yet does not know her own genetic status or that of her affected sister. She has received some counseling in the past regarding options related to infertility but is likely not aware of the full ramifications of the multiple decision pathways that are now before her. Dr. Bowers must carefully explore these various options with Mrs. Castle and her family—all in the context of what is likely booked as a 15-30 minute appointment.
Initial Counseling
At the outset of the appointment, ahead of a full discussion, Dr. Bowers is encouraged to proceed with a serum pregnancy test, which we will presume is positive and significant enough to warrant an ultrasound. Mrs. Castle may decline the ultrasound, yet the information afforded would be essential. Her difficulties in conceiving are likely related to her advanced age of 41 years, which puts the pregnancy at increased risk of aneuploidy and miscarriage. Thus, to the extent possible at this early stage, confirming a pregnancy should be the first step. While this process could require multiple appointments, Dr. Bowers, recognizing the time constraints at issue, should expedite this process as best she can.
Assuming a pregnancy that appears to be proceeding normally at approximately 8-10 weeks, we would encourage Dr. Bowers to consult the timeline below and to carefully discuss each of the possible pathways with Mrs. Castle, assuming she wishes to continue her pregnancy (see figure 1). Authors have previously described this very situation as an “existential crisis” for the patient and family [1]. Dr. Bowers might also feel the weight of this existential crisis, given the difficulty of addressing these complex decisions and their far-reaching implications under significant time constraints. A comprehensive discussion of counseling and possible testing is difficult to achieve even ahead of pregnancy in the setting of assisted reproduction, in vitro fertilization (IVF), and preimplantation genetic diagnosis (PGD), which we discuss more fully below. In fact, it might be nearly impossible when a patient is faced with an ongoing pregnancy and no knowledge of familial genetic risk. Thus, our primary advice to Dr. Bowers is to seek help from an interdisciplinary team. A single clinician, no matter how skilled, is unlikely to be able to provide the counsel and support that Mrs. Castle and her family need as they grapple with these challenging decisions.
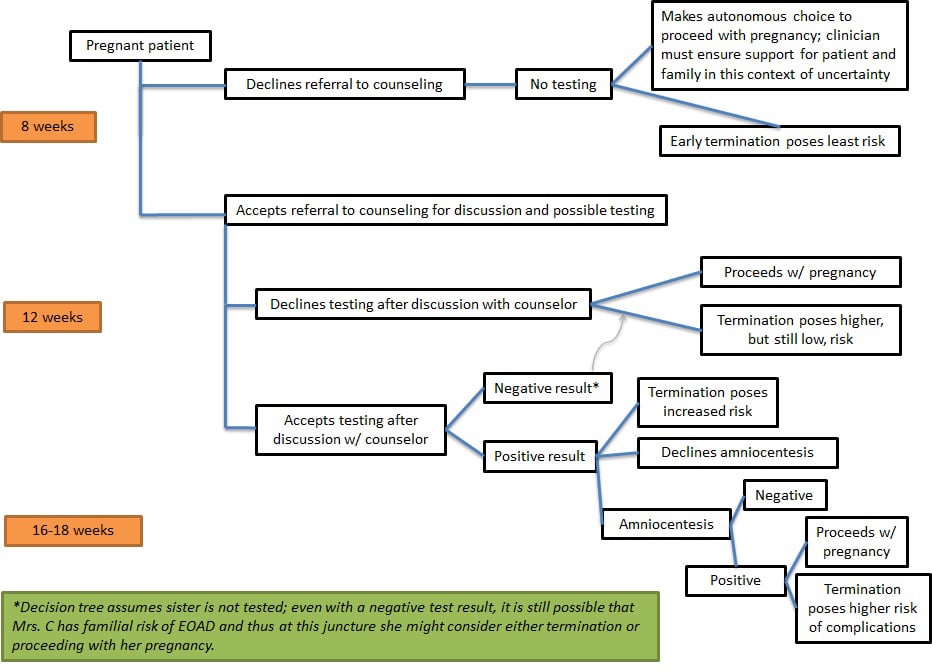
Figure 1. Decision tree and timeline
(Click image to enlarge.)
But first Dr. Bowers should pose to Mrs. Castle a simple question: Does she wish to continue this pregnancy? The likely answer would be yes—she has been trying to conceive for over a year. However, one can imagine a patient who, faced with this existential crisis, would choose at the outset to terminate. Perhaps Mrs. Castle has been avoiding full consideration of what her sister’s diagnosis and her own symptoms might mean for herself and a potential child. Confronted starkly with these prospects, she might find that continuing with a pregnancy is simply not possible for her or her family, even without genetic test results. A full discussion of the ethics of abortion is beyond the scope of this paper. Suffice it to say, should Mrs. Castle request termination at any stage of this process (as indicated in figure 1), we will assume her request would be granted.
The next step in our timeline would be to encourage Mrs. Castle and her family, including her sister and her sister’s family, to involve genetic counselors and neurologists in her care. To prepare Mrs. Castle for her meetings with these clinicians, Dr. Bowers should discuss the potential downstream consequences of these consults. For example, Dr. Bowers should explain that testing would take weeks, which would push related decisions further along the timeline—and further into Mrs. Castle’s pregnancy. Dr. Bowers should provide a general overview of testing and the options that would arise along each decision pathway. She should also flag the possibility of fetal testing by amniocentesis and the difficulties Mrs. Castle could face in considering this possibility. The details of this discussion are addressed more fully below.
Given the complexities involved, this level of counseling would take hours and might be beyond the scope of what an obstetrician-gynecologist would feel comfortable addressing in a clinic visit. Yet merely referring Mrs. Castle to a genetic counselor without preparing her would not be appropriate. Depending on what consultants are available to Dr. Bowers, involvement of a maternal fetal medicine specialist could be helpful in facilitating her care.
In short, Dr. Bowers should approach this situation as a counseling emergency of sorts, given the timeline, and should involve all those who can assist this family as quickly as possible. What follows is a detailed discussion of the salient points to be addressed in the counseling process.
Overview of Genetic Testing in the Setting of EOAD
Mrs. Castle has a strong family history of Alzheimer’s disease (AD), which represents 60-70 percent of dementia cases worldwide [2]. Like other forms of dementia, AD causes progressive deterioration in cognitive, emotional, and social functioning [3].
AD is categorized as early onset (i.e., before age 65) or late onset (LOAD) [4]. The early-onset form of the disease (EOAD) represents 10 percent of all AD cases [5]. Strikingly, the inherited genetic contribution to EOAD is estimated to be 92-100 percent [6]. Strictly speaking, EOAD is not synonymous with autosomal dominant AD, but because autosomal dominant AD is found almost exclusively in families with EOAD, this article will use these terms interchangeably [4].
Genetic testing for EOAD detects any of the three known autosomal dominant mutations in the amyloid precursor protein (APP) and presenilins 1 and 2 (PSEN1 and PSEN2) genes [7]. Mutations in the APP and PSEN1genes are completely penetrant, so people with the mutant allele will develop AD if they live a normal lifespan [4]. A person with either of these mutations has a 50 percent chance of passing the mutant allele on to each child [8]. By contrast, mutations in PSEN2 are 95 percent penetrant [4]. Although mutations in any of the three known EOAD genes are causative, these mutations only account for 5-10 percent of all cases of EOAD [5]. In short, a person can receive a negative test result for these mutations and remain significantly at risk for developing EOAD.
In addition to mutations in the three EOAD genes, the &egr;4 allele of the apolipoprotein E (APOE) gene increases a person’s susceptibility to developing both EOAD and LOAD [4]. However, as predictive APOEgenotyping is neither recommended nor routinely practiced, it falls beyond the scope of this article [4].
Benefits of Genetic Counseling
Genetic counseling is an interactive process focused on educating the patient about the inheritance, progression, and management of genetic disease [9]. A genetic counselor would discuss Mrs. Castle’s eligibility for testing and serve as a neutral source of information and a partner in reflection as she considers the available options in light of their risks and her own values [10].
Per current medical guidelines, people who have symptoms of EOAD, at least one family member with EOAD, and/or a family member with one of the known causative mutations, are eligible for genetic testing [4]. As Mrs. Castle has two family members with EOAD and is herself apparently symptomatic, she is a candidate for genetic testing. Given the clinical ramifications and emotional burden of genetic testing, the guidelines advise that testing proceed only with neurological evaluation and with genetic counseling both pre- and posttest [4]. Simply put, this process is very time intensive.
To increase the likelihood of obtaining an informative result, the guidelines further recommend that a living, affected family member—in this case, Mrs. Castle’s sister—undergo testing for a known causative mutation first [4]. In addition, Mrs. Castle’s sister and her family should be advised to consult with a neurologist for possible panel testing to clarify her diagnosis and ascertain whether other factors might be contributing to her dementia. This information stands to benefit the entire family, including Mrs. Castle. Unfortunately, it is not clear that the sister has the capacity to consent to testing, that she would agree to be tested if she had capacity, or that consent could be obtained quickly enough to have any bearing on decisions that Mrs. Castle might make regarding this pregnancy.
It is critical to note here that without first testing Mrs. Castle’s sister, a negative test result for Mrs. Castle affords little information about her risks of developing EOAD (see figure 2). Insofar as mutations in the three identified EOAD genes account for only a small percentage of all cases of EOAD, in this scenario, Mrs. Castle could still face a significant risk of developing the disease [5]. Assuming that information about the sister’s genetic status is not available, Mrs. Castle must be supported in making a difficult decision about whether to be tested herself so as to make a decision about her pregnancy.
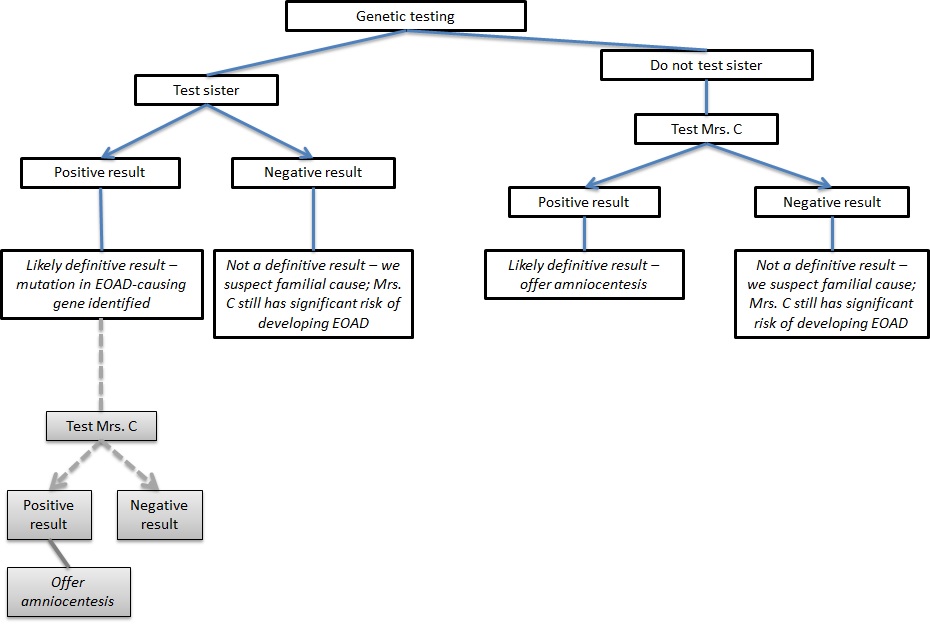
Figure 2. Genetic testing decision tree
(Click image to enlarge.)
Given the pressures involved in these emotional decisions, Dr. Bowers should emphasize ahead of referral that genetic counseling offers significant psychotherapeutic and educational value and does not commit Mrs. Castle to genetic testing [11]. Mrs. Castle should expect genetic counseling to contextualize her risk and provide an informative, nondirective discussion of the implications of the disease. She should also expect that the counselor will review the decisions that she will have to make and again review their possible consequences, as did Dr. Bowers in her initial appointment.
While we might take genetic testing to be a value-neutral mode of providing information, the utilization of test results in clinical decision making could prompt vexing concerns about truly autonomous choice. More specifically, scholars of disability theory raise ethical concerns that genetic counseling and testing could be implicitly directive in a way that diminishes the value of persons who fall outside constructions of able-bodiedness [12]. One safeguard against this implicit directedness might involve including insights from the lived experiences of persons with the disease. As disease is not strictly physiological but unfolds within a social context, genetic counseling should provide information about both the clinical and social dimensions of living with disease [12-14]. Toward that end, the genetic counselor should discuss with Mrs. Castle that both she and her child, if affected, could live as many as 64 asymptomatic years before the onset of EOAD symptoms, and that the experience of dementia, like that of other conditions, does not ipso facto preclude a rich and meaningful human existence [15].
Preconception Testing
As mentioned multiple times in this discussion, the time constraints associated with this case are far from ideal. Mrs. Castle faces both the stress of pregnancy and the prospect of learning powerful information that could affect her own future and that of her fetus. Ideally, the clinical and ethical deliberation concerning genetic testing for Mrs. Castle would have preceded conception. This sequence of events would allow for a more extensive discussion of options, including in vitro fertilization (IVF) with preimplantation genetic diagnosis (PGD) and subsequent transfer of unaffected euploid embryos. Although costly, this option might be available to a patient who wished to transfer unaffected embryos without knowing her own genetic status [1]. Certainly, this option is not without complexities. Clinicians must consider the patient’s exercise of autonomy alongside the breadth of parental latitude granted to the patient in making decisions for a future child. The tensions inherent in these decisions are similar to those present in the setting of possible amniocentesis, discussed below. Yet, at the very least, this option affords the family and clinicians time to more meaningfully explore questions related to quality of life that necessarily impact decision making in this context.
Post Counseling
Proceeding along our timeline (see figure 1), following counseling, Mrs. Castle must now decide whether or not to accept testing. Despite the limitations discussed above, even assuming that her sister has not been tested, Mrs. Castle should consider genetic testing for herself. If Mrs. Castle tests positive and wishes to know whether the fetus is affected, she will have to undergo prenatal testing in the second trimester by amniocentesis, discussed further below [16].
Assuming that her symptoms indicate EOAD, Mrs. Castle might decline testing on the grounds that it would reveal what she already believes to be true. Mrs. Castle might surmise that she is likely affected but might wish to remain in a state of ignorance, thereby shielding herself from the knowledge that she might develop a progressive and potentially devastating disease. In this scenario, one could conceive of the possibility of proceeding to amniocentesis to determine if the fetus is affected. However, we would argue that this option should be discouraged. Certainly, this is an area of debate, as some might posit that respect for Mrs. Castle’s autonomy demands she be afforded this option. We would argue that the potential for a definitive result here is exceedingly small, as there is no knowledge of the genetic susceptibility in this family. Consequently, Mrs. Castle is in a position to learn either a devastating or a functionally useless result. In the setting of a positive result, she would learn that she and her fetus, along with her sister and other family members, are affected by a mutant allele. Alternatively, in the setting of a negative result, which is much more likely, she would learn very little relevant information.
Should Mrs. Castle instead opt to undergo testing and be found to carry an EOAD-causing mutation, Dr. Bowers and the genetic counselor’s first priority would be to work with the other involved health care professionals to continue to tailor care to Mrs. Castle’s needs. Even if she received a negative result, in consultation with her neurologist, she could be faced in the short term with a diagnosis of EOAD and thus the counseling that follows testing would still apply. Mrs. Castle will need to consider how best to adapt to her changes in functioning, establish a sound financial plan, and so on. She might also believe that her family has an important role—beyond the caregiving roles they are likely to assume—in helping her to grapple with these decisions. Insofar as her priorities, values, and preferences are shaped through these relationships, Mrs. Castle will be best off considering her options through conversation and reflection with those most important to her.
In this interpersonal context, Mrs. Castle will need to think about how to anticipate a shift—not necessarily a downgrade—in her quality of life with the onset of AD. To be sure, evaluating quality of life is a thorny issue in the context of disease, and Alzheimer’s dementia is no exception. Conversations between Mrs. Castle and each of her caregivers should address considerations unique to the experience of dementia and how they should be weighed in her reflections [17]. The declines in cognitive and social functioning that accompany dementia raise significant challenges to our ordinary thinking about a person’s future wishes. How might Mrs. Castle plan for the years ahead when her future self is so discontinuous with her current circumstances that extrapolation about her future interests seems impossible? For example, Mrs. Castle might not currently get much enjoyment from watching sitcoms on television or eating an ice cream cone, but it is difficult, if not altogether impossible, to know whether she might take pleasure in these activities after the onset of her dementia. Does she want what is most important to her now to remain an important reference point at a future time when perhaps her interests—and certainly her neurological functioning—have changed considerably? Such questions about the continuity of the self—and the continuity of one’s future wishes and judgments about an acceptable quality of life—merit far deeper discussion than we can delve into here. Nevertheless, meaningful reflection on these important and complex questions will undoubtedly suffer under the tight timetable of an existing pregnancy.
Fetal Testing
If she receives a positive test result, Mrs. Castle will then have to quickly decide whether to learn the genetic status of her fetus. Prior to pursuing amniocentesis, Mrs. Castle must consider how she will respond if the test yields a positive result. In this context of prenatal genetic diagnosis, Mrs. Castle’s judgments about her future child’s quality of life generate even more complex questions. Does Mrs. Castle have the ethical authority to determine that her future child’s quality of life would be so diminished by EOAD that it would be in the child’s own interest not to exist at all? Or does such a judgment fall beyond the purview of Mrs. Castle’s decision-making authority, especially when it is not clear how the future child might evaluate his or her own quality of life as he or she experiences the onset of dementia symptoms as an adult? As Mrs. Castle and her family consider these questions, the involved clinicians should serve as partners in reflection.
In addition to these ethical concerns, Mrs. Castle must consider the slight risk of miscarriage that amniocentesis carries. The procedure-related risk of miscarriage for amniocentesis and chorionic villus sampling (CVS) is approximately 0.1 percent and 0.2 percent, respectively [18]. If Mrs. Castle does not anticipate that the prenatal diagnosis will influence her decision about continuing the pregnancy—that is, if the procedure offers no foreseeable benefit—we find no ethical warrant for posing this risk to the fetus. However, if Mrs. Castle plans to use the prenatal diagnosis in making her decision, we argue that testing might be worth the risk. In such cases, the clinician’s role should center on accurately communicating the procedure-related risks and then allowing the patient to determine the course of action most consistent with her and her family’s values [19].
Conclusion
The field of genetic diagnosis is expanding rapidly. As evidenced by this case, patients and clinicians will have to engage more and more directly with difficult ethical dilemmas surrounding genetic testing.
In our discussion above, we have laid out the various decisions confronting Mrs. Castle, her family, Dr. Bowers, and the clinicians who will necessarily be involved in her care. From a clinical perspective, we find most daunting the prospect of the first meeting, during which so much information must be conveyed. To proceed with any first step in our decision tree by truly autonomous decision making, Mrs. Castle must understand the downstream implications of each step. Achieving such a level of understanding amidst the anxiety that Mrs. Castle is experiencing seems nearly impossible. Recognizing genuine understanding as an important element of reproductive choice, we emphasize that education of patients about the decisions involved in genetic testing should precede pregnancy. Yet it is unlikely this education could easily be attained for most patients.
Ethical quandaries abound at each node of the decision tree, and, as is frequently the case in reproductive ethics, they center on questions of autonomy and its proper scope. Early involvement of an interdisciplinary team to address these questions is essential in supporting our patients as they make these difficult decisions.
References
- de Die-Smulders CE, de Wert G, Liebaers I, Tibben A, Evers-Kiebooms G. Reproductive options for prospective parents in families with Huntington’s disease: clinical, psychological, and ethical reflections. Hum Reprod Update. 2013;19(3):304-315.
-
World Health Organization; Alzheimer’s Disease International. Dementia: A Public Health Priority. Geneva, Switzerland: World Health Organization; 2012.
http://apps.who.int/iris/bitstream/10665/75263/1/9789241564458_
eng.pdf?ua=1. Accessed February 2, 2017. - McKhann G, Knopman S, Chertkow H, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):263-269.
- Goldman J, Hahn SE, Catania JW, et al. American College of Medical Genetics; the National Society of Genetic Counselors. Genetic counseling and testing for Alzheimer disease: joint practice guidelines of the American College of Medical Genetics and the National Society of Genetic Counselors. Genet Med. 2011;13(6):597-605.
- Cacace R, Sleegers K, Van Broeckhoven C. Molecular genetics of early-onset Alzheimer’s disease revisited. Alzheimers Dement. 2016;12(6):733-748.
- Wingo TS, Lah JJ, Levey AI, et al. Autosomal recessive causes likely in early-onset Alzheimer disease. Arch Neurol. 2012;69(1):59-64.
- Chouraki V, Reitz C, Maury F, et al. International Genomics of Alzheimer’s Project. Evaluation of a genetic risk score to improve risk prediction for Alzheimer’s disease. J Alzheimers Dis. 2016;53(3):921-932.
- Bird TD. Genetic aspects of Alzheimer disease. Genet Med. 2008;10(4):231-239.
- Resta R, Biesecker B, Bennett R, et al. National Society of Genetic Counselors’ Definition Task Force. A new definition of genetic counseling: National Society of Genetic Counselors’ task force report. J Genet Couns. 2006;15(2):77-83.
- Elwyn G, Gray J, Clarke A. Shared decision making and non-directiveness in genetic counselling. J Med Genet. 2000;37(2):135-138.
- Inglis A, Koehn D, McGillivray B, Stewart SE, Austin J. Evaluating a unique, specialist psychiatric genetic counseling clinic: uptake and impact. Clin Genet. 2015;87(3):218-224.
- Madeo A, Biesecker BB, Brasington C, Erby LH, Peters KF. The relationship between the genetic counseling profession and the disability community: a commentary. Am J Med Genet A. 2011;155A(8):1777-1785.
- Ahmed S, Bryant L, Hewison J. “Balance” is in the eye of the beholder: providing information to support informed choices in antenatal screening via Antenatal Screening Web Resource. Health Expect. 2007;10(4):309-320.
- Koch T. Disability and difference: balancing social and physical constructions. J Med Ethics. 2001;27(6):370-376.
-
Newell C. The social nature of disability, disease, and genetics: a response to Gillam, Persson, Holtug, Draper and Chadwick. J Med Ethics. 1999;25:172-175.
- Chasen ST, Kalish RB, Chervenak FA. Gestational age at abortion: the impact of first-trimester risk assessment for aneuploidy. Am J Obstet Gynecol. 2006;195(3):839-842.
-
Ready RE, Ott BR. Quality of Life measures for dementia. Health Qual Life Outcomes. 2003;1:11. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC155631/. Accessed June 1, 2017.
- Akolekar R, Beta J, Picciarelli G, Ogilvie C, D’Antonio F. Procedure-related risk of miscarriage following amniocentesis and chorionic villus sampling: a systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2015;45(1):16-26.
-
Lehmann LS. How can we improve amniocentesis decision-making? Isr J Health Policy Res. 2016;5:4. https://ijhpr.biomedcentral.com/articles/10.1186/s13584-016-0060-0. Accessed June 1, 2017.




