Case
As pediatrician Dr. Carson completes her physical exam of Alex, a healthy 15-year-old girl, she senses that Alex has something else on her mind. “Alex, I’m happy to say that you’re in perfect health. Do you have any more questions for me before we bring your mother back into the room?”
“My boyfriend and I are thinking about having sex,” Alex responds. “I’ve never had sex before.” Dr. Carson asks a few more questions, reviews safe sex practice and provides prophylactic condoms. “You should also know that the CDC recommends the HPV vaccine to girls starting at age 11 to prevent infections that may one day cause cancer.” Dr. Carson continues to describe the vaccine’s efficacy, prevalence, and known side effects. Alex decides she would like to receive the vaccine.
Dr. Carson asks Alex’s mother to re-enter the room and begins to discuss the recommendation for the HPV vaccine. Alex’s mother objects. “The HPV vaccine is mandated here in Virginia for 11-year-old girls. I would have chosen to opt out of the mandate because I don’t want my daughter to act irresponsibly or suffer unknown side effects.”
Alex responds angrily. “I’ll come back to Dr. Carson’s office alone, or get the vaccine at Downtown Family Planning,” she says, referring to a Title-X clinic near where they live in Richmond. Dr. Carson believes that, although physicians can legally provide both reproductive health care and STI diagnosis and treatment to a competent minor, they cannot legally administer the HPV vaccine without parental consent.
Commentary
Pediatricians, family physicians, and ob-gyns who routinely care for adolescent girls will be familiar with many of the ethical and practical challenges that Dr. Carson faces in this office visit: confidentiality, access to health care, consent, and conflicts between public health policy and personal health beliefs. Walking through this vignette, with stops for discussion along the way, we review some of these common challenges while addressing unique features added by the HPV vaccine.
“Do you have any more questions for me before we bring your mother back into the room?”
By providing Alex an opportunity to discuss matters privately, Dr. Carson follows a recommended practice for gaining the trust of adolescent patients—a practice that is supported by ethical principles, legal analyses, and research findings [1, 2]. “Time alone” has been shown to significantly increase the likelihood that adolescents will discuss sensitive health matters and is generally supported by parents and appreciated by patients [1-3].
“My boyfriend and I are thinking about having sex,” Alex responds. “I’ve never had sex before.” Dr. Carson asks a few more questions, reviews safe sex practice and provides prophylactic condoms.
Alex has responded by bringing up a sensitive health issue, offering Dr. Carson an important opportunity to provide information and preventive health recommendations.
Because many adolescent patients see their primary care physicians infrequently and at those times for acute matters only, opportunities to provide preventive health care can be few and far between [2, 4]. In our clinical case scenario, Alex has come to Dr. Carson for a physical exam. We’d expect Dr. Carson to allow “time alone” for Alex to bring up sensitive matters, but we’d also expect Dr. Carson to ask about sexual health in a way that sets the stage for current and future discussions. Note that as the conversation progresses, Dr. Carson misses an opportunity to find out what Alex has discussed with her mother and how they should bring her mother into the discussion. Recognizing and negotiating this tripartite relationship is critical to best practices for adolescents [1, 2].
Reproductive health care and diagnosis and treatment of sexually transmitted infections (STIs) are important topics when adolescents go to the doctor. However, the types of services that can be offered without parental consent or knowledge vary considerably from state to state [5]. Some states allow minors to consent to all forms of medical treatment as long as the minor meets specified criteria set by law (e.g., particular age, married, high school graduate). There are also specific statutory exemptions, varying by state, that allow minors to receive certain kinds of medical services without parental consent or knowledge (e.g., family planning, testing and treatment for STIs, mental health care, and/or substance abuse counseling).
So far, Dr. Carson is likely to be within the ethical and legal bounds of adolescent care and confidentiality in most states, although practitioners should take care to know the law of the state in which they practice. The law can contain unexpected gaps and incongruities. For example, in Virginia, where Dr. Carson practices and Alex lives, minors can consent to the kinds of medical services listed above in parentheses. At the same time, Virginia parents have a right to obtain their child’s health records unless the disclosure of those records would be reasonably likely to cause substantial harm to the minor or another person [6]. As a result, in some circumstances parental permission and knowledge might be avoided initially, but the minor patient’s privacy and confidentiality may not be entirely protected.
In the clinical scenario under discussion the possibility of inadvertent disclosure should prompt Dr. Carson to take extra care when recording information in Alex’s medical record—better to document the conversation with Alex in a general way than to reveal what Alex confided about her boyfriend. Physicians should also be mindful of the fact that regardless of laws that might protect minors’ confidentiality against disclosure to parents, “explanation of benefits” in insurance statements sent to parents may specify the medical services for which a claim is made [1, 7]. While this is not an issue with respect to advice about safe sex practices and the provision of condoms, it would be relevant if reimbursement were sought for other services such as laboratory testing for sexually transmitted infections.
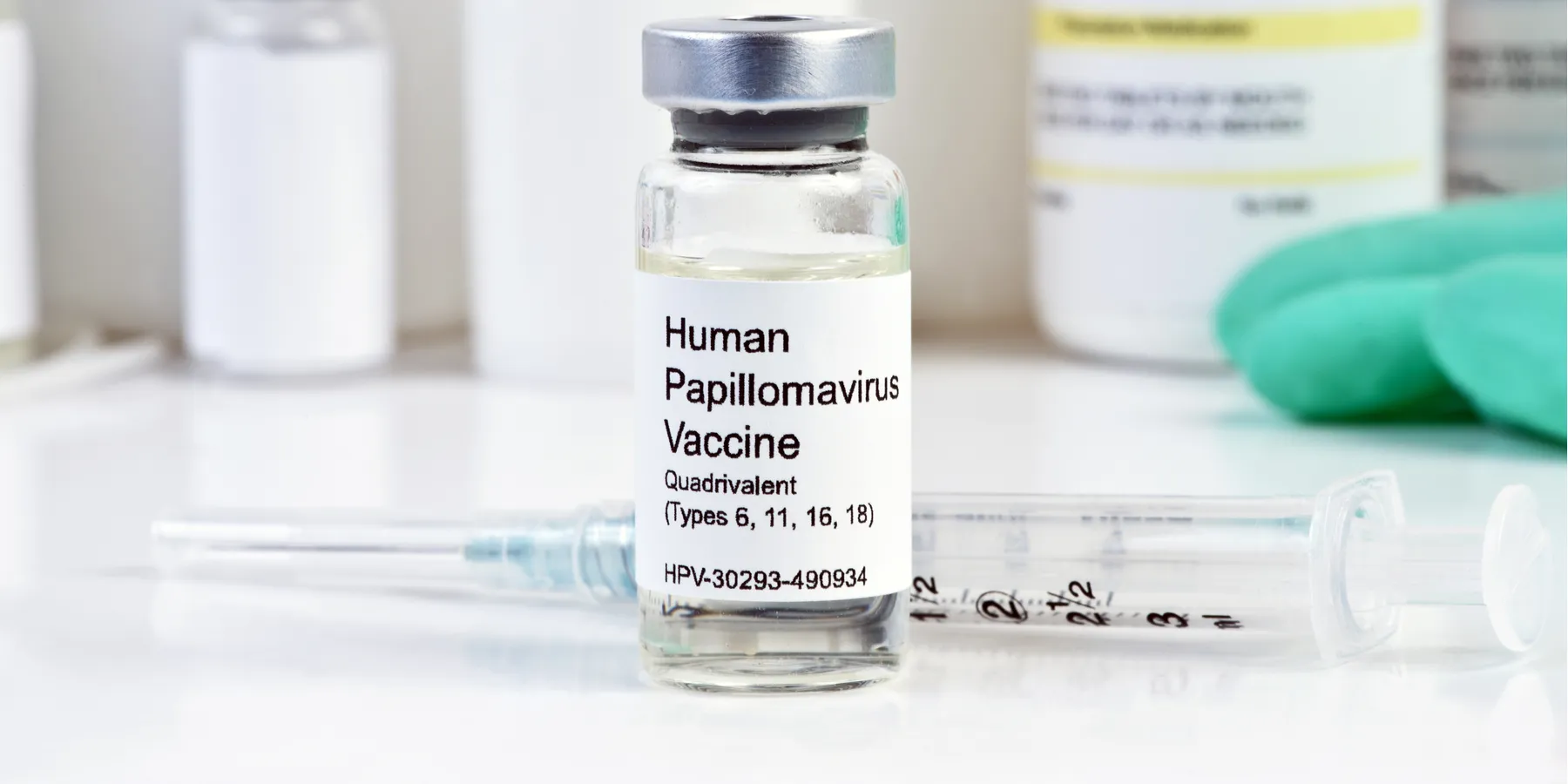
“You should also know that the CDC recommends the HPV vaccine to girls at age 11 to prevent infections that may one day cause cancer.”
Chronic infection with high-risk human papillomavirus (HPV) causes almost all cancer of the cervix. Condoms are only partially effective in preventing HPV transmission. Two vaccines are available: Gardasil, a quadrivalent vaccine, targets HPV strains 6, 11, 16, and 18; Cervarix, a bivalent vaccine, targets strains 16 and 18. Large clinical trials as well as epidemiological studies have demonstrated vaccine effectiveness in preventing genital warts, precancerous lesions, throat cancer, and anal cancer as well as cervical cancer [8-10].
HPV vaccines are more effective if administered before sexual debut and therefore before HPV infection. While the median age of first sexual intercourse in the U.S. is 17 years, 13 percent of girls engage in sexual activity before age 15 and 4 percent before age 13 [11]. In 2007 the CDC recommended that the three-dose schedule of the HPV vaccine be given to girls aged 11-12, and catch-up vaccination to girls and women aged 13-26 years.
Until recently male transmission of HPV was not factored into national vaccine policy. In 2009 Gardasil was approved for boys aged 9-26 years, and in October 2011 the CDC added routine HPV vaccination for boys aged 11-12 and a catch-up vaccine for boys and men aged 13-21. Some experts would give the catch-up vaccine to men as old as 26. Vaccinating boys and men may reduce rates of anal and throat cancer in men and also reduce cancer in women by reducing HPV transmission [12]
Dr. Carson provides an important service in discussing the HPV vaccine with Alex, although one has to wonder why the vaccine was not discussed during earlier visits with Alex and her parents, either as a part of a “well adolescent” visit or as part of routine review of recommended vaccines. Such reviews are an integral part of adolescent health care, as patients may need to “catch up” on vaccinations usually administered to children and also need to be informed about new vaccines. For example, three “new” vaccines have been licensed since 2005 and are recommended for 11-12-year-olds: HPV; MCV4 (the first meningococcal conjugate vaccine); and Tdap (the new tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccine) [13].
Earlier doctor-patient-parent discussion in the context of routine vaccinations and preventive health care may have tempered HPV vaccine’s association with adolescent sexual activity in general and Alex’s sexuality in particular. But Dr. Carson’s timing is not unusual. Despite CDC recommendations, physicians are more likely to recommend HPV vaccination for 13-15-year-olds than for 11-12-year-olds [11, 14]. The perceived need to discuss sexuality with patients before offering the HPV vaccine appears to serve as a barrier both to recommendation by physicians and acceptance by parents [11]. This may be an artifact of the introductory phase of this vaccine. Just as with hepatitis B, if HPV vaccine is part of a panel of vaccines offered at the same time, the perceived link to sexuality may decrease.
Alex’s mother objects, “The HPV vaccine is mandated here in Virginia for 11-year-old girls. I would have chosen to opt-out of the mandate because I don’t want my daughter to act irresponsibly or suffer unknown side effects.”
Virginia is one of two jurisdictions (along with Washington, D.C.) to mandate HPV vaccine prior to middle school entry. The mandate, enacted in 2008, did not affect Alex, but, as her mother’s statement suggests, the mandate does not obviate the need for parental permission, and the grounds on which parents can opt out of the mandate are generous. All state mandatory vaccination laws allow parents to opt out for legitimate medical reasons, many allow opt-outs on the basis of religious beliefs, and some for philosophical or personal beliefs [15]. The option to decline the HPV vaccination in Virginia is quite broad: parents can refuse the vaccine for any reason after having reviewed materials describing the link between HPV and cervical cancer [16].
Such parental reluctance is not unusual. A national survey of pediatricians and family physicians conducted in 2008, 18 months after HPV vaccine was licensed, found that over half of the physicians surveyed reported that at least one-fourth of parents of 11- to 12-year-old patients refused (defined as “outright refusal without plans for future vaccination”) or deferred (defined as “postponing vaccination with the intention of considering it in the future”) HPV vaccine. Refusals and deferrals were less frequent for 13- to 15-year olds [11]. While few physicians think that vaccination encourages earlier or riskier sexual behavior, almost half reported that parents were concerned about this.
Even though vaccine safety is a concern shared by physicians and parents, these concerns have lessened with time [4, 11]. Nevertheless, parents still worry that HPV vaccine is not safe enough. By 2008 few physicians reported concerns about HPV vaccine safety, but over one-quarter of surveyed physicians reported that parental worries about safety remained a barrier to vaccine acceptance [11]. The CDC website on vaccine safety includes up-to-date information about the HPV vaccines [17]. As of June 2011, over 35 million doses of Gardasil had been distributed, and postmarketing surveillance data attest that Gardasil remains generally safe and effective. Because adolescents are prone to fainting or orthostasis after injections, with risk of falls and injury, both the FDA and the CDC recommend lying down for 15 minutes after receiving the vaccine.
Education helps to overcome parents’ reluctance to consent to the HPV vaccine, especially education that highlights the reduced risk of cervical cancer after vaccination [11]. Physician recommendation increases immunization acceptance in general and the same is true for HPV vaccine [11, 18]. A recent study shows that a strong physician recommendation of the HPV vaccine results in four times the rate of parental support [19]. One of many toolkits to help educate patients and their parents about the vaccine is available at http://www.hpvvaccineproject.org/hpv.php?page=providers_involved. The American Academy of Pediatrics (AAP) website also has helpful information about ways to respond to various reasons for refusing vaccination, as well as suggestions for documenting the discussion and coding the reason why a vaccine was not administered.
Alex responds angrily. “I’ll come back to Dr. Carson’s office alone, or get the vaccine at Downtown Family Planning,” she says, referring to a Title-X clinic near where they live in Richmond.
As discussed above, the kinds of medical treatment that adolescents can consent to without their parents is determined by state law. In Virginia Dr. Carson is most likely correct in her assessment that she cannot legally give Alex the vaccine without parental consent. While minors in Virginia can consent to medical services “required in case of birth control, pregnancy or family planning,” as well as to services that test or treat sexually transmitted or other reportable diseases, the statutes do not explicitly cover prevention of sexually transmitted diseases. The law in other states may, although the language in the Virginia statutes is fairly typical. When these statutes were adopted the existence of a vaccine to protect against a sexually transmitted infection was not anticipated, so the fact that the statutes do not contain specific exclusion or inclusion of such preventive measures is not surprising [20]. California recently responded to this omission and passed specific legislation to allow minors to consent to HPV vaccination [21].
If Dr. Carson cannot give Alex the vaccine without parental consent, Alex will not have any better luck receiving the vaccine by coming back to Dr. Carson’s office alone. But, what if Alex goes to a nearby family planning clinic supported by federal Title X funds? Such a clinic may provide the vaccine, although that is not guaranteed, as services are not uniform across Title X clinics, and the vaccine is expensive, about $120 per injection for a series of three injections. In addition, while Title X clinics are legally required to safeguard the confidentiality of minors seeking services there, the scope of the services they can provide without parental consent may be limited by the same state laws that apply in Dr. Carson’s office. This is an area of the law that is both unclear and evolving [2, 22].
Dr. Carson finds herself in an uncomfortable and unfortunate situation. She should let both Alex and her mother know that she is not able to provide the vaccination without parental consent. It is possible that a strong recommendation by Dr. Carson will persuade Alex’s mother to allow Alex to receive the vaccine, if not during this visit, then a later one. Before forcing the issue and taking a strong position in favor of the vaccine, Dr. Carson may need to meet with Alex and her mother separately, and confidentially, followed by a three-way conversation that looks for common ground and promotes trust. Time usually helps. Dr. Carson can provide Alex and her mother some literature on the vaccine and suggest that they give it some more thought and come back to discuss at a return appointment, which would optimally be scheduled before they leave the office.
References
- Ford C, English A, Sigman G. Confidential health care for adolescents: position paper for the society for adolescent medicine. J Adolesc Health. 2004;35(2):160-167.
- Berlan ED, Bravender T. Confidentiality, consent, and caring for the adolescent patient. Curr Opin Pediatr. 2009;21(4):450-456.
- McKee MD, Rubin SE, Campos G, O’Sullivan LF. Challenges of providing confidential care to adolescents in urban primary care: clinician perspectives. Ann Fam Med. 2011;9(1):37-43.
- Kimmel SR. Adolescent vaccination: physician challenges and solutions. J Adolesc Health. 2010;46(2 Suppl 2):s16-s23.
- English A, Shaw FE, McCauley MM, Fishbein DB. Working Group on Legislation, Vaccination, and Adolescent Health. Legal basis of consent for health care and vaccination for adolescents. Pediatrics. 2008;121(Suppl 1):S85-S87.
-
Va Code section 54.1-2969.
-
Gold RB. Unintended consequences: how insurance processes inadvertently abrogate patient confidentiality. Guttmacher Policy Rev. 2009;12(4):12-16. http://www.guttmacher.org/pubs/gpr/12/4/gpr120412.html. Accessed December 18, 2011.
-
Centers for Disease Control and Prevention (CDC). FDA licensure of bivalent human papillomavirus (HPV2, Cervarix) for use in females and updated HPV vaccine recommendations from the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep. 2010;59(20):626.
-
Markowitz LE, Dunne EF, Saraiya M, et al. Quadrivalent human papillomavirus vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep. 2007;56(RR-2):1.
- Marra F, Cloutier K, Oteng B, Marra C, Ogilvie G. Effectiveness and cost-effectiveness of human papillomavirus vaccine: a systematic review. Pharmacoeconomics. 2009;27(2):127-147.
- Daley MF, Crane LA, Markowitz LE, et al. Human papillomavirus vaccination practices: a survey of US physicians 18 months after licensure. Pediatrics. 2010;126(3):425-433.
- Peres J. Who should get the HPV vaccine? Usage expands amid debate. J Natl Cancer Inst. 2010;102(12):838-840.
- Kaplan DW. Barriers and potential solutions to increasing immunization rates in adolescents. J Adolesc Health. 2010;46(2 Suppl 2):s24-s33.
- Vadaparampil ST, Kahn JA, Salmon D, et al. Missed clinical opportunities: provider recommendations for HPV vaccination for 11-12 year old girls are limited. Vaccine. 2011;29(47):8634-8641.
-
National Conference of State Legislatures. States with religious and philosophical exemptions from school immunization requirements; 2011. http://www.ncsl.org/default.aspx?tabid=14376. Accessed November 8, 2011.
-
Va Code section 32.1-46(B).
-
Centers for Disease Control and Prevention. Frequently asked questions about HPV safety. http://www.cdc.gov/vaccinesafety/Vaccines/HPV/hpv_faqs.html. Accessed November 8, 2011.
- Dorell CG, Yankey D, Santibanez TA, Markowitz LE. Human papillomavirus vaccination series initiation and completion, 2008-2009. Pediatrics. 2011;128(5):830-839.
- Rosenthal SL, Weiss TW, Zimet GD, Ma L, Good MB, Vichnin MD. Predictors of HPV vaccine uptake among women aged 19-26: importance of a physician’s recommendation. Vaccine. 2011;29(5):890-895.
- Farrell RM, Rome ES. Adolescents’ access and consent to the human papillomavirus vaccine: a critical aspect for immunization success. Pediatrics. 2007;120(2):434-437.
-
Cal Assembly Bill 499 (2011-2012).
-
Gold RB. Challenges and opportunities for U.S. family planning clinics in providing the HPV vaccine. Guttmacher Policy Rev. 2007;10(3). http://www.guttmacher.org/pubs/gpr/10/3/gpr100308.html. Accessed December 18, 2011.