Cancer drugs are big business. Worldwide sales are projected to reach $25 billion in 2006 and to increase to almost $50 billion by 2010 [1]. This represents a startling growth in a segment of the drug industry once shunned by major pharmaceutical manufacturers as too high-risk and unprofitable. While a few drug companies, notably Bristol-Myers Squibb (BMS) and Pharmatalia, made significant profits on cancer drugs between 1970 and 1990 when the first effective combination therapies came into common practice, the turning point in this industrial segment occurred in 1992 with the approval of Bristol-Myers Squibb's paclitaxel, which became a multibillion-dollar-per-year product by 1998.
To understand our current concerns with cancer drug costs and their potential effect on medical care financing and access, one needs to be familiar with the paclitaxel experience. The story of paclitaxel's discovery and commercial development reflects both the lack of interest that industry had in cancer drugs at that time and the sudden emergence of drug cost as a social justice issue.
In 1964 Monroe Wall and associates, working at the Research Triangle Institute under a National Cancer Institute (NCI) contract, isolated the active compound in paclitaxel from the bark of the common yew tree [2]. Its tortuous development, complicated by difficulties in material procurement, compound purification and formulation, delayed its entry into clinical trials until 1983, and its efficacy in treating ovarian cancer was not demonstrated until 1987 [3]. Because of the need to procure large amounts of plant material for its isolation and the tendency of the necessary solvent (Cremophor EL) to cause hypersensitivity reactions, there was little commercial interest in the compound. When NCI announced an open competition for clinical development of the compound in 1990, only four companies responded. Two of the applicants were small firms, unprepared for the task of drug production and clinical development. A third was a foreign company that already held rights to a competitor compound in the same class. BMS was the only major U.S. company to apply for the development rights and won the contract in 1991. Shortly thereafter, the drug's effectiveness against breast cancer became apparent, and it emerged as a blockbuster.
Congressional involvement
Pricing of the drug immediately became a concern for the U.S. Congress [4]. Because it had been discovered and developed under government contracts with the NCI, members of Congress scrutinized the price set by BMS. The NCI had urged BMS to set a price consistent with that of competitive compounds in the field of ovarian cancer. NCI directors believed that the production costs, limited range of uses and novelty of the compound justified a cost per treatment of approximately $2,000, and the BMS price was consistent with this target price. NCI also wanted to assure that patients could get the drug regardless of their insurance status, a concern to which BMS responded by setting up a program of free distribution to indigent patients. But members of Congress castigated BMS at open hearings, pointing out the government's key role in the drug's discovery and deploring the profit BMS was making. An unstated, but recurring theme in these hearings was the plight of cancer patients who had no choice but to pay or seek insurer reimbursement for this uniquely effective medication. Even though the drug was shown to confer a survival advantage in multiple forms of cancer, most notably breast and ovarian, some countries, including those in the United Kingdom, refused to grant approval based on a cost-benefit analysis. Parenthetically, the U.K. National Health Service continues to deny use of other expensive new drugs, such as bortezomib, on the same basis.
During this tumultuous period of paclitaxel marketing, when the U.S. Congress and the public first directly confronted the pharmaceutical industry over the cost of life-saving cancer drugs, it became apparent that the public had no other option but to pay the price. Federally mandated price controls were openly discussed at Senate hearings, but rejected as impractical (what is a fair price?) and potentially fatal to the rapidly growing biotechnology industry. No one—neither physicians nor the patients in need—could place an appropriate dollar value on the worth of one year of human life [5].
Challenges for pricing drugs in the 21st century
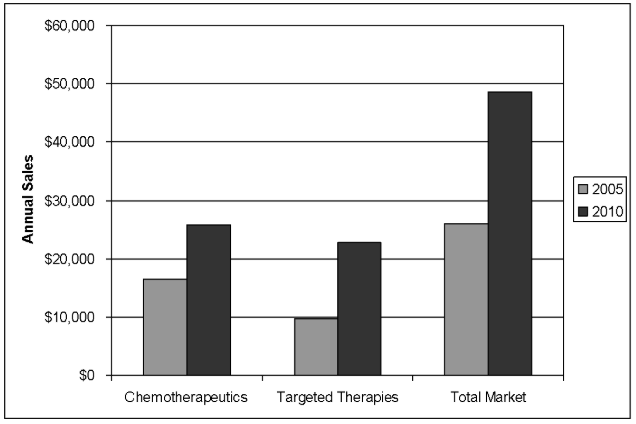
The paclitaxel experience set the stage for the dilemma of cancer drug pricing that is now playing out on a much larger scale. Since the mid-1990s the biotechnology industry has made major contributions to cancer treatment with targeted therapies—agents that have specific molecular targets and which, alone, are not toxic to cells at standard doses. (Traditional chemotherapy agents are cytotoxins that cause cell death at standard doses.) Targeted therapies include monoclonal antibodies such as bevacizumab (Avastin) and trastuzumab (Herceptin) from Genentech; selective small molecule tyrosine kinase inhibitors such as erlotinib (Tarceva) from OSI pharmaceuticals/Genentech Inc.; and multi-targeted kinase inhibitors such as sorafenib (Nexavar; Onyx Pharmaceuticals/Bayer AG) and sunitinib (Sutent; Pfizer Inc.). The antibodies have achieved annual sales in excess of $1 billion each, and their potential for further expansion seems unlimited (figure 1).
Figure 1. Estimated Worldwide Market for Oncology Drugs by Class (in millions) in 2005 and 2010 (Data are from S.G. Cowen and Company; Reference 1).
The cost of these medications ranges from approximately $3,000 per treatment cycle for the small molecules to $7,000-$10,000 per treatment cycle for the antibodies [6]. One antibody in particular, cetuximab (Erbitux; Imclone Systems Inc/BMS), an EGFR (epidermal growth factor receptor) inhibitor indicated for treatment of colorectal and head and neck cancer, has attracted significant publicity because of its high cost and low response rates. While each of the companies has established mechanisms that provide assistance for uninsured or indigent patients, the high cost of these new medications has attracted considerable attention in the medical and lay press [7]. How can it be justified?
The risk-reward ratio for companies engaged in cancer drug discovery and development remains unfavorable. The cost begins with a major investment in basic research, often heavily supplemented by NCI and other grants, followed by extensive preclinical evaluations and clinical trials involving hundreds of patients and many years of effort. If one takes into account the expenses associated with failed drugs, the industry spends approximately $1 billion for each compound that reaches the market [8]. Obviously, for the individual successful compound and its company, the cost is significantly less, but there is no doubt that this is not an industry for the faint-hearted or for those with shallow pockets. No more than 7 percent of cancer compounds that enter clinical trials end up reaching the market.
While several hundred biotech companies are now engaged in cancer drug discovery and development, the number of new drugs approved each year remains in the single digits, and most companies ultimately fail to earn a profit. Certain classes of promising compounds, such as vaccines and cell cycle inhibitors, in which multiple companies have invested hugely, have not yet produced a single approved drug. Recent public and congressional concerns about the need for post-marketing surveillance to ensure safety would further increase the cost of drug development.
Pretrial systems for predicting clinical success, based on mouse models of human disease or human tumor cell lines, have largely failed. When breakthrough drugs such as imatinib (Gleevec; Novartis AG) for chronic myelogenous leukemia do succeed, their period of uniqueness is often brief, as competitors quickly produce new and perhaps better drugs, such as dasatinib (Sprycel; BMS), for the same target illness [9]. And, finally, the period of patent protection, typically 20 years from the time of patent filing, is too brief, considering the time—10 years on average—spent in development and the fact that the pharmaceutical industry must reinvest up to 30 percent of its profits in new drug research.
Forces that may reduce the cost of cancer drugs
While talk of price controls continues in Congress, other factors are likely to mitigate pricing. The first is competition. A new antibody, panitumumab (Vectibix; Amgen Inc.), an effective EGFR inhibitor, has entered the market in competition with cetuximab (Erbitux) and costs less. A number of small molecules are in the late stage of development and are being groomed to compete with the most expensive drugs, namely the monoclonal antibodies. There are differences between the antibodies and their small molecule competitors (e.g., site of action, target access, pharmacokinetics, etc.), so a lot of comparative development remains to be done. Nonetheless, the small compounds, traditionally priced below the antibodies, will, if approved, most likely drive down the cost of cancer care. Orally administered small molecules have the additional attractive feature of not requiring a hospital visit and thus obviate the cost of intravenous infusion, a major economic benefit for the health care system. As prescription drugs, however, they are not always covered completely by the insurance of those who need them; that is, they can fall into Medicare's proverbial "donut hole" of noncoverage.
A second force for reducing the cost of care will be improvements in patient selection for therapy. With few exceptions, cancer drugs are presently used in settings in which only a fraction of patients will benefit. Cetuximab, which is among the most costly antibodies, produces responses in 10-15 percent of patients with chemotherapy-resistant colorectal cancer. It may benefit a larger subset of patients when given in combination with irinotecan, but there is no test currently available to identify the responsive subset of patients.
Through the use of molecular diagnostics (biomarkers), it may be possible to improve the response rates and eliminate the needless expense of "shotgun" therapy. Examples of successful patient selection include the use of imatinib in chronic myelogenous leukemia (CML), in which all patients have tumors with translocations involving the same gene. Further, CML patients who develop resistance to imatinib harbor further mutations, most of which are sensitive to dasatinib [9]. Molecular analysis clearly has a role in creating a treatment plan for these resistant patients. The experience with EGFR inhibitors such as erlotinib and gefitinib has yielded a strong correlation between receptor mutations and responsiveness, a relationship that could lead to up-front or adjuvant use of these drugs in selected patients [10].
The NCI and Food and Drug Administration (FDA) have jointly endorsed the development of biomarkers for drug selection in cancer and have outlined an ambitious research effort. At the same time, however, the FDA is tightening its oversight of "home brew" diagnostic tests, asking for stronger prospective validation of assays. In the past, reimbursement for these tests was determined by the regional Medicare carrier and by individual insurers. Depending on the standard to which these tests will be held, the development of molecular diagnostics for cancer could encounter significant regulatory delays in the future.
Finally, the government possesses a strong weapon in bargaining for lower prices of cancer drugs. It is a major purchaser of pharmaceuticals through the Veterans Administration system and sets reimbursement rates for medical procedures and services in the Medicare program. Congress is threatening to become more involved in issues of drug pricing, using its bully pulpit—congressional hearings—to expose excessive profits. Through legislative action, it could ask for a cost-benefit analysis as part of Medicare reimbursement policy and could extend patent life as a reward for corporate programs that expand free access to drugs, ensuring that all patients will benefit from the federally funded research that underlies virtually all of these new discoveries.
In conclusion, the high cost of cancer drugs tests corporate responsiveness to public needs, while challenging scientific innovation and the potential of the competitive marketplace to control prices. The federal government will surely exercise indirect influence over pricing through its ability to expose the issue in congressional hearings and through the multiple points of intersection of the executive branch and industry. Meanwhile scientific progress is driving the cost of cancer care ever upward. This progress is saving lives, but there have to be limits. At this point, no one has a clear idea what these limits might be.
References
-
S.G. Cowen and Company. Pharmaceutical Therapeutic Categories Outlook: Comprehensive Study. New York, NY: Cowen and Company; 2006:503.
- Wani MC, Taylor HL, Wall ME, Coggon P, McPhail AT. Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J Am Chem Soc. 1971;93(9):2325-2327.
- Rowinsky EK, Donehower RC. Paclitaxel (Taxol). N Engl J Med. 1995;332(15):1004-1014.
-
Goodman J, Walsh V. The Story of Taxol: Nature and Politics in the Pursuit of an Anti-Cancer Drug. Cambridge, UK: Cambridge University Press; 2001.
-
Nadler E, Eckert B, Neumann PJ. Do oncologists believe new cancer drugs offer good value? Oncologist. 2006;11(2):90-95.
- Schrag D. The price tag on progress—chemotherapy for colorectal cancer. N Engl J Med. 2004;351(4):317-319.
-
Berenson A. A cancer drug shows promise, at a price that many can't pay. New York Times. February 15, 2006:A1, C2. Available at: http://www.nytimes.com/2006/02/15/business/15drug.html?ex=1297659600&en=bc6aaaf25acffa44&ei=5088. Accessed December 12, 2006.
-
Brock DW. How much is more life worth? Hastings Cent Rep. 2006;36(3):17-19.
- Kantarjian H, Jabbour E, Grimley J, Kirkpatrick P. Dasatinib. Nat Rev Drug Discov. 2006;5(9):717-718.
- Lynch TJ, Bell DW, Sordella R, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004;350(21):2129-2139.