Abstract
Overprescription of antibiotics in cases in which bacterial infection is clinically uncertain contributes to increased prevalence of multidrug-resistant bacteria. Ethically, merits and drawbacks of stricter prescription practice oversight should be weighed against risks of untreatable bacterial infections to patients and communities. This article considers how to balance this set of ideas and values.
Resistance as an Ethical Problem
The rise of antibacterial resistance worldwide, combined with the projected increase in morbidity and mortality that patients will suffer in consequence,1 has prompted many clinicians and ethicists to rethink their approach to antibiotic use. The problem of antibacterial resistance originates from complex and multifactorial challenges that extend well beyond the hospital or clinical setting. However, overuse of antibiotics within clinical encounters remains a major contributor to the development of drug-resistant bacteria.2 Even appropriate use of antibiotics contributes to a minute but collectively gradual loss of their effectiveness for patients, communities, and members of future generations. Stewardship measures pertaining to antibiotic prescribing practices and policies need to address underlying ethical tensions, including the medical need of an individual patient and the minimization of antibacterial resistance for the community.
Uncertainty
In order to characterize the need of an individual patient as it pertains to antibiotic prescribing, it helps to stratify a patient’s risk of bacterial infection into 1 of 3 categories: (1) clinical suspicion of bacterial infection is near or at 100%, such that it is ethically and medically mandatory to prescribe antibiotics; (2) there is no clinical suspicion of bacterial infection—it is ethically and medically mandatory to not prescribe antibiotics; and (3) clinical suspicion of bacterial infection is uncertain, and it is ethically permissible and medically justifiable to prescribe antibiotics. It is unlikely that anyone would say the first category is problematic. Although antibiotics are inappropriately prescribed for viral infections and noninfectious conditions,3,4 the second category described, with risk of bacterial infection known with confidence to be nonexistent, is not particularly ethically challenging, either. So why do many patients with viral illnesses leave their clinic appointment with antibiotic prescriptions in hand?5
When clinicians view antibiotic prescribing through the lens of uncertainty, there is a tendency to favor minimizing the risk of potential bacterial infection. Even if a viral infection is more likely, the clinician’s duty to prevent harm seemingly supports providing antibiotics because the risk of bacterial infection is not zero. This response is likely both a result of, and a direct contributor to, a culture of “zero tolerance” for avoidable infection-related harm that is reinforced by defensive antibiotic prescribing6 and cognitive biases that influence decision making. These biases include commission bias (favoring action over inaction), hyperbolic discounting (favoring small, immediate gains over long-term benefits or reduction in harm), and optimism bias (overestimating potential benefits and underestimating risks).7 An example of these biases influencing both patient expectations and clinician practices is the association between antibiotic prescribing volume and both patient demand and higher patient satisfaction scores,8,9 regardless of whether the antibiotic was medically necessary or not.
Prevention
Two of the prevailing ethical frameworks within antibiotic stewardship are a cost-benefit approach, most often represented as utilitarianism, and contractualism. While both frameworks are related to the central claim made here, which is that antibiotic stewardship should include a patient-centered, harm-reduction approach, a detailed discussion of different ethical theories as they pertain to antibiotic stewardship as a collective issue falls outside the scope of this work. Utilitarians10 would claim that clinicians are expected to weigh risks and benefits in terms of outcomes as they relate to everyone, not just the individual patient. In this instance, antibiotics should only be prescribed when the expected benefits to everyone outweigh the expected costs. Contractualists, discussed in the context of antibacterial resistance by Michael Millar,11,12 would state that the clinician should only prescribe antibiotics when guided by principles that no one could reasonably reject. Millar defines one such principle as prescribing antibiotics to prevent “substantial risk of irretrievable harm” in patients or their contacts and introduces the idea that the level of acceptable risk should be above the level of risk we consent to in our daily lives, such as in driving a car. Using antibiotics when the risk of harm is comparable to or lower than these activities should be avoided for the purpose of protecting the community from further antibacterial resistance.12 Both the utilitarian and contractualist approaches require the clinician to account for the well-being of people other than their patient, whether these people compose the community or future generations.
However, both approaches undervalue the special relationship clinicians share with their patient. It seems both practically and ethically appropriate that clinicians have particular obligations to prevent harm to their patients rather than to prevent harm to everyone, even if the latter consideration should not be disregarded completely. Although Millar’s suggested risk threshold for antibiotic use accounts for antibiotic-associated risk to the patient, it is difficult to empirically conceptualize and does not seem well suited to the context of clinical care with all its pragmatic complexities, including diagnostic uncertainty and clinical and epidemiological factors that increase or decrease confidence in a bacterial diagnosis on a patient-by-patient basis. Furthermore, clinicians have traditionally expressed extremely low tolerance for introducing even a small risk of avoidable harm to their patients,13 especially in the form of underdiagnosis or undertreatment. The risk of introducing or enhancing antibacterial resistance is among the least important factors they account for when making antibiotic prescribing decisions.14
Centering Patients
Unless we can appeal to clinicians’ duty to prevent any avoidable harm to their patients, there is little reason to think their gatekeeper-like influence over antibiotic prescribing will dramatically change. Utilitarian and contractualist approaches are unsatisfactory and unlikely to sway clinicians to change their antibiotic prescribing practices. However, discussions of trade-offs in antibiotic prescribing frequently rely on the assumption that antibiotics are inherently beneficial and that harm only occurs in their absence. Yet, as clinicians know, antibiotics can be a direct cause of adverse medical outcomes. The expected utility to an individual patient of receiving antibiotics can be lower than the expected utility of not receiving antibiotics, even when the risk of potential bacterial infection is more than zero. A clinical example of this scenario is when antibiotics are prescribed for patients with COVID-19, which is discussed in detail below. Reframing the justification for limiting antibiotic prescribing to one that focuses on harm reduction for a given patient—including risk of the potential for both bacterial infection and antibiotic-related adverse events—appeals to a clinician’s duty to prevent harm while also indirectly benefiting the community by reducing low-utility antibiotic prescriptions.
Take the example of COVID-19, a viral infection. Differences between an isolated COVID-19 infection and a bacterial co-infection can be difficult to distinguish clinically. By several months into the pandemic, studies showed a wide range of bacterial co-infection rates in hospitalized COVID-19 patients, but a meta-analysis of multiple studies found an average co-infection rate of 7%.15 This rate decreased to 1% to 3% for mild and moderate COVID-19 cases.16 Yet, as many as 50% to 90% of patients admitted for COVID-19 during the first year of the pandemic received antibiotics.15,17,18 While a proportion of these prescriptions were appropriate for select patients at increased risk of having a bacterial co-infection, such as patients requiring admission to intensive care,19,20,21,22,23 the incidence of co-infection among these small but high-risk populations still did not approach the overall rates of empiric antibiotic use.
In order to compare the relative risks and benefits of prescribing or not prescribing antibiotics for an individual COVID-19 patient, we can conduct a thought experiment to estimate the expected utility of each therapeutic decision, as shown in Table 1. These expected utilities are admittedly subjective quantifications of potential utility selected to reflect the perspective of a clinician faced with the decision to give or not give antibiotics to a patient with COVID-19. For simplicity, this example assumes that the antibiotic prescribed is appropriate for co-infection. The utility of prescribing antibiotics when a bacterial co-infection is present is high (let us say +100), while the utility of not providing antibiotics for a bacterial co-infection is inversely proportional, if not worse (-150). In giving antibiotics for a purely viral process, there is an appreciable risk of negative utility in the form of an antibiotic adverse event (-20)—which is based on a study showing that 20% of hospital patients who receive an antibiotic experience an adverse antibiotic-related event24—and a small positive utility in not giving antibiotics in the form of prevention of future antibacterial-resistant infection in the patient (+5). As shown in the right-most column, which multiplies this subjective expected utility by the rate of bacterial co-infections (7%) to generate overall expected utility, even abstaining from prescribing antibiotics for potential bacterial co-infection among routine COVID-19 infection cases comes with a degree of risk; it is in the patient’s best interest not to receive antibiotics empirically, without needing to directly account for any related community benefit.

Some may disagree with this formulation because it relies on subjective quantifications of utility that vary based on clinician preferences, level of clinical uncertainty, or amount of information available. As an alternative, we can compare antibiotic-associated adverse outcomes among COVID-19 cases with and without bacterial co-infection. As mentioned previously, 20% of patients who receive antibiotics in the hospital experience at least one adverse drug reaction.24 Extrapolating from this statistic, roughly 1 of every 5 COVID-19 patients receiving antibiotics without bacterial co-infection will suffer from an antibiotic-related adverse outcome without any relevant health gains (see Figure). Comparing these patients as a proportion of all patients without bacterial co-infection to those receiving antibiotics with bacterial co-infection, it can be concluded that antibiotics cause avoidable harm to COVID-19 inpatients without bacterial co-infection at a rate (eg, between 8 and 16 patients of every 93 patients, or between 9% to 17% based on the Figure) that is higher than the 7% of patients with COVID-19 who benefit from antibiotics because a clinician accurately diagnoses and treats their bacterial co-infection.
Figure. Prevalence of Bacterial Co-Infection, Antibiotic Use, and Antibiotic-Associated Adverse Events in Hospitalized Patients With Routine COVID-19 Infection
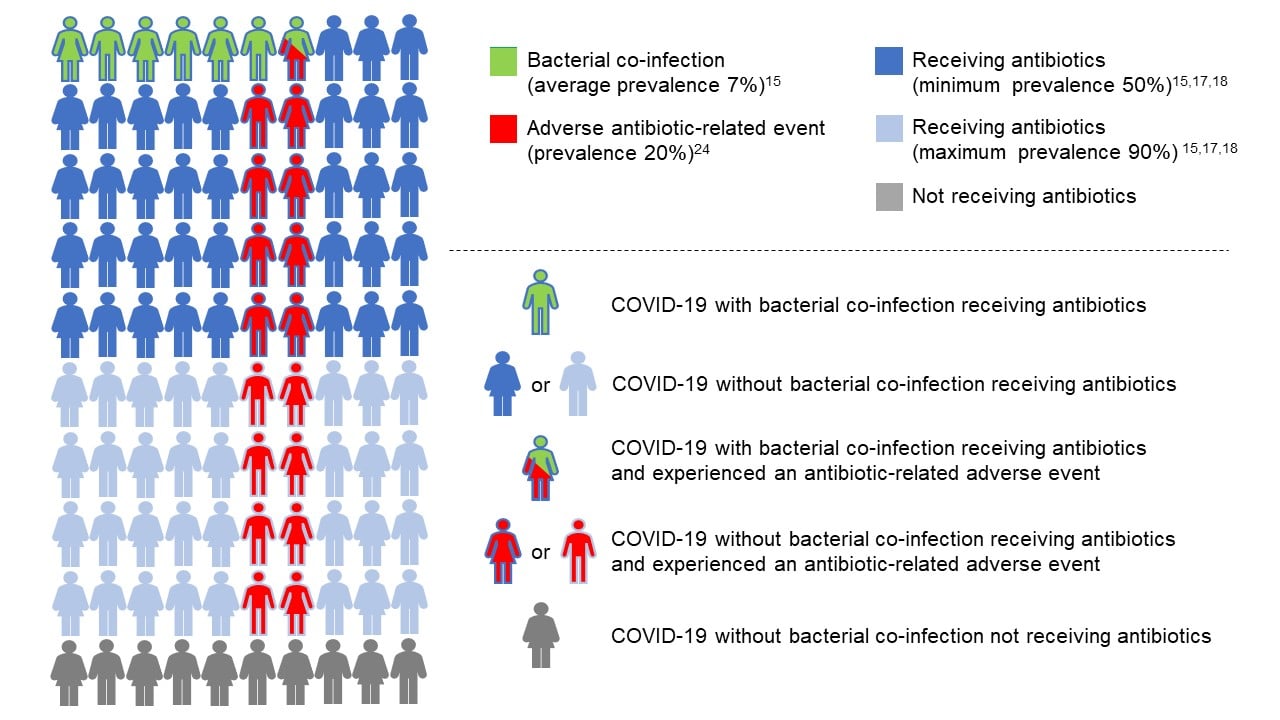
Green symbols with a blue outline represent patients receiving appropriate treatment. While a proportion of these patients will still have an antibiotic-associated adverse event (red and green symbol with blue outline), this risk is tolerable if not justifiable due to the need for antibiotics to prevent bacterial infection-related harm. Conversely, solid blue symbols represent patients receiving overtreatment with antibiotics in the absence of bacterial co-infection. A proportion of these patients will have one or more antibiotic-associated adverse events (red symbols with blue outline). It seems difficult to justify incurring this harm, given the lack of any benefit.
Table 2 enumerates other commonly encountered clinical scenarios in which antibiotics or antifungals are often prescribed despite not being indicated in most cases. However, for other situations that also fall under the category of uncertainty, the pretest probability of bacterial infection is higher, such as patients who are critically ill or who have immunocompromising conditions, which can justifiably tip the calculus in favor of prescribing antibiotics.

One limitation of the expected utility framework is that it does not address certain factors, such as patient preferences and expectations, that influence how a clinician weighs relative risk, benefit, and utility in antibiotic decision making. This shortcoming highlights one of the many areas in which a robust antimicrobial stewardship program can—by enacting processes such as antibiotic approvals, preset antibiotic durations, and individual clinician audits—prevent actions that result in overprescribing antibiotics, underestimating potential harm from side effects, or prescribing antibiotics that are too broad, are too narrow, or are used for too long of a duration, given the indication. These informed risk-benefit calculations can be discussed with patients in order to reach a decision that both patient and clinician feel is consistent with the patient’s best interest.
Conclusion
Addressing the rise in antibacterial resistance via antibiotic stewardship is an emerging priority for health systems and clinicians. There is certainly no straightforward solution, and there remains a distinct need for normative deliberation and empirically supported methods to reduce individual clinician prescribing practices that contribute to antibacterial resistance. Suggestions to increase, even slightly, the potential risk of avoidable harm to a patient for the sake of community benefit are incompatible with how a clinician’s duty to prevent harm is ingrained in professional norms. In health care systems where access to antibiotics is via prescription from a clinician, there is potential advantage in reframing restricting antibiotic prescribing as focusing on the patient and in revisiting how we conceptualize patient harm. When there is a degree of uncertainty about a bacterial diagnosis, a utility calculus or harm-reduction approach can be useful for antibiotic decision making. In cases in which expected utility favors not prescribing antibiotics, there is both a direct benefit to the patient by reducing avoidable harm from antibiotics and an indirect benefit to the community by decreasing one of the major selective pressures that promote antibacterial resistance.
References
-
Murray CJL, Ikuta KS, Sharara F, et al; Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399(10325):629-655.
-
Antimicrobial resistance. World Health Organization. November 21, 2023. Accessed February 29, 2024. https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance
-
Kronman MP, Gerber JS, Grundmeier RW, et al. Reducing antibiotic prescribing in primary care for respiratory illness. Pediatrics. 2020;146(3):e20200038.
- White AT, Clark CM, Sellick JA, Mergenhagen KA. Antibiotic stewardship targets in the outpatient setting. Am J Infect Control. 2019;47(8):858-863.
- Hersh AL, King LM, Shapiro DJ, Hicks LA, Fleming-Dutra KE. Unnecessary antibiotic prescribing in US ambulatory care settings, 2010-2015. Clin Infect Dis. 2021;72(1):133-137.
- Charani E, Ahmad R, Rawson TM, Castro-Sanchèz E, Tarrant C, Holmes AH. The differences in antibiotic decision-making between acute surgical and acute medical teams: an ethnographic study of culture and team dynamics. Clin Infect Dis. 2019;69(1):12-20.
-
Langford BJ, Daneman N, Leung V, Langford DJ. Cognitive bias: how understanding its impact on antibiotic prescribing decisions can help advance antimicrobial stewardship. JAC Antimicrob Resist. 2020;2(4):dlaa107.
- Ashworth M, White P, Jongsma H, Schofield P, Armstrong D. Antibiotic prescribing and patient satisfaction in primary care in England: cross-sectional analysis of national patient survey data and prescribing data. Br J Gen Pract. 2016;66(642):e40-e46.
- Kohut MR, Keller SC, Linder JA, et al. The inconvincible patient: how clinicians perceive demand for antibiotics in the outpatient setting. Fam Pract. 2020;37(2):276-282.
- Johnson T. A trade-off: antimicrobial resistance and COVID-19. Bioethics. 2021;35(9):947-955.
- Millar M. “Zero tolerance” of avoidable infection in the English National Health Service: avoiding the redistribution of burdens. Public Health Ethics. 2013;6(1):50-59.
- Millar M. Constraining the use of antibiotics: applying Scanlon’s contractualism. J Med Ethics. 2012;38(8):465-469.
- Fiscella K, Franks P, Zwanziger J, Mooney C, Sorbero M, Williams GC. Risk aversion and costs: a comparison of family physicians and general internists. J Fam Pract. 2000;49(1):12-17.
- Metlay JP, Shea JA, Crossette LB, Asch DA. Tensions in antibiotic prescribing: pitting social concerns against the interests of individual patients. J Gen Intern Med. 2002;17(2):87-94.
- Lansbury L, Lim B, Baskaran V, Lim WS. Co-infections in people with COVID-19: a systematic review and meta-analysis. J Infect. 2020;81(2):266-275.
- Wang L, Amin AK, Khanna P, et al. An observational cohort study of bacterial co-infection and implications for empirical antibiotic therapy in patients presenting with COVID-19 to hospitals in North West London. J Antimicrob Chemother. 2021;76(3):796-803.
-
Garcia-Vidal C, Sanjuan G, Moreno-García E, et al; COVID-19 Researchers Group. Incidence of co-infections and superinfections in hospitalized patients with COVID-19: a retrospective cohort study. Clin Microbiol Infect. 2021;27(1):83-88.
- Vaughn VM, Gandhi TN, Petty LA, et al. Empiric antibacterial therapy and community-onset bacterial coinfection in patients hospitalized with coronavirus disease 2019 (COVID-19): a multi-hospital cohort study. Clin Infect Dis. 2021;72(10):e533-e541.
-
Pickens CO, Gao CA, Cuttica MJ, et al; NU COVID Investigators. Bacterial superinfection pneumonia in patients mechanically ventilated for COVID-19 pneumonia. Am J Respir Crit Care Med. 2021;204(8):921-932.
-
Baskaran V, Lawrence H, Lansbury LE, et al. Co-infection in critically ill patients with COVID-19: an observational cohort study from England. J Med Microbiol. 2021;70(4):001350.
-
Adler H, Ball R, Fisher M, Mortimer K, Vardhan MS. Low rate of bacterial co-infection in patients with COVID-19. Lancet Microbe. 2020;1(2):e62.
-
Karaba SM, Jones G, Helsel T, et al. Prevalence of co-infection at the time of hospital admission in COVID-19 patients, a multicenter study. Open Forum Infect Dis. 2020;8(1):ofaa578.
-
Musuuza JS, Watson L, Parmasad V, Putman-Buehler N, Christensen L, Safdar N. Prevalence and outcomes of co-infection and superinfection with SARS-CoV-2 and other pathogens: a systematic review and meta-analysis. PLoS One. 2021;16(5):e0251170.
- Tamma PD, Avdic E, Li DX, Dzintars K, Cosgrove SE. Association of adverse events with antibiotic use in hospitalized patients. JAMA Intern Med. 2017;177(9):1308-1315.
- Zervos M, Meunier F. Fluconazole (Diflucan): a review. Int J Antimicrob Agents. 1993;3(3):147-170.