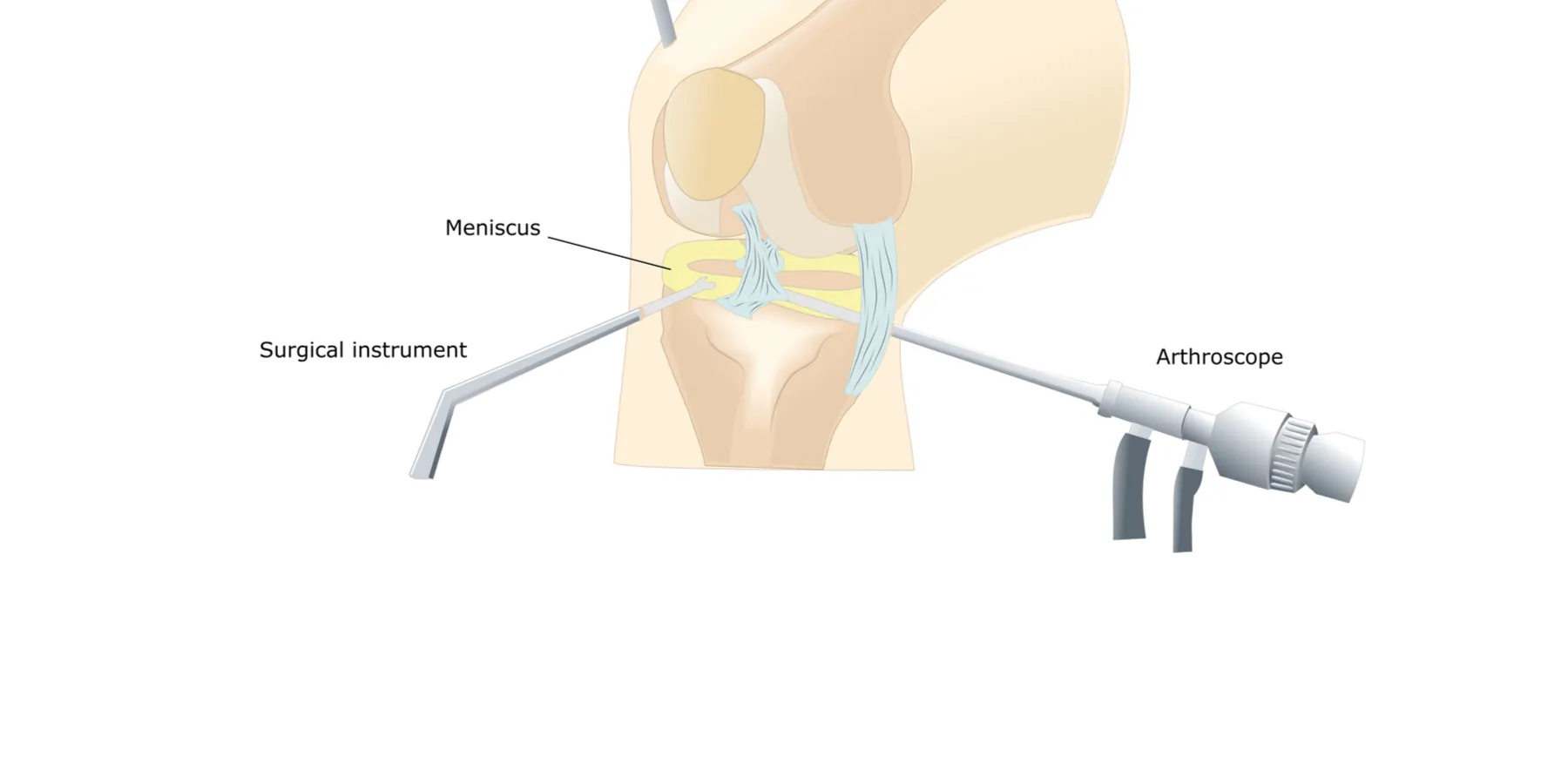
Case
Dr. Janus is an orthopedic surgeon who specializes in arthroscopic surgery and keeps current with medical research in his area. His goal is not only to provide the best care but also the most effective treatment at the lowest cost for his patients. Lately, he has treated many elderly patients with osteoarthritis of the knee and has noticed that this type of surgery is often a financial burden for his patients. Although the surgery is quite expensive, at $4,500 per knee, patients continue to request it, hoping to improve their mobility. In reviewing the current literature, Dr. Janus learns that the type of arthroscopic knee surgery he has been performing routinely (arthroscopic lavage and debridement) was shown in two different studies to provide no benefit over placebo; in fact, a strong placebo effect has been suggested. Dr. Janus feels that the patients he operated upon usually did improve, but he cannot rule out the placebo effect.
Mr. Marcus is a 70-year-old retiree living on his limited pension. He suffers from advanced osteoarthritis in his knees and would do anything to be able to walk more easily on his own. He says to Dr. Janus, “My best friend had this surgery last month, and he’s already getting around the golf course great. He says this surgery was the best thing he ever did for himself. I sure would love to be able to keep up with him now. What do you say, Doc, can you do this surgery for me?”
Commentary
The scenario described in this vignette is frequently seen by orthopedic surgeons. Arthroscopy of the knee is one of the most common orthopedic procedures performed in the United States [1-3] and has resulted in significant reduction in patient pain and improvement in function. However, some conditions and symptoms, such as pain osteoarthritis in the absence of mechanical symptoms such as locking, catching, and giving-way, have not been found to improve following arthroscopy [4-12]. Although roughly 650,000 arthroscopic procedures for osteoarthritis of the knee at a cost of approximately $5000 each [1] are performed annually in the United States [13], evidence available on the efficacy of this procedure is inconclusive [4-6, 14]. Most clinical series [4-12, 15, 16] have shown success rates between 50 and 75 percent. It is this lack of success that prompted Moseley et al. [13] in 2002 to investigate the benefit of arthroscopic surgery in a group of older adults suffering from osteoarthritis of the knee.
The study included 180 patients from the Houston Veteran’s Affairs Medical Center, 75 years of age and younger, with knee osteoarthritis who reported at least moderate pain despite maximal medical treatment for at least 6 months. They were randomized to three study arms: arthroscopic debridement, arthroscopic lavage, or a sham operation consisting of three 1-cm portal incisions without penetration of the joint capsule. The primary outcome measure was knee pain 2 years after surgery. The authors found no significant difference between treatment arms of the study, thus concluding that routine arthroscopic lavage, debridement, or both were no better than the sham procedure.
Critics of this study point out that flexion weight-bearing radiographs were apparently not done to fully discern the degree of cartilage wear, nor were X-rays taken of the entire lower extremity to assess the mechanical axis of the limb; the study population was largely male and may not be representative of the general population; the degree to which the patients experienced mechanical knee symptoms was not well-described; and the authors used the Knee-Specific Pain Scale as their primary end point even though this is a non-validated measurement that was “created for this study.” Therefore, there was harsh criticism from many orthopedic surgeons following the publication of this controversial study.
This study, however, does not lead us to recommend a sham operation to persons suffering from osteoarthritis of the knee. Rather, it suggests that routine knee arthroscopy for osteoarthritis of the knee is not clinically beneficial to any significant degree in those patients who resemble those who participated in the Moseley trial. Considering the financial burden this procedure puts on an already stressed U.S. medical system, unless other studies offer a contrary conclusion, orthopedic surgeons should exhaust other, less expensive, less risky, and more effective treatment options for patients with osteoarthritis of the knee.
Use of Placebo in Clinical Trials
This case raises questions about the potential clinical benefit and ethical ramifications of placebo-controlled trials in surgery. “Equipoise” is considered a central ethical element to consideration of placebo controls. Equipoise refers to uncertainty about which arm of a trial may have greater benefits or harms. In its most basic form, equipoise represents a state of genuine and credible doubt among knowledgeable researchers about the relative therapeutic merits of some set of interventions that target a specific medical condition; to many it represents a necessary condition for ethically acceptable human-subjects research [17].
It is widely accepted that the purpose of evidence-based medicine is to implement medical therapies as “proven” by multicenter randomized controlled trials (RCTs). The current gold standard of evidence is the double-blind RCT, in which the therapy of interest is compared to the accepted treatment or to a placebo. The term “placebo” is commonly used to describe any substance or procedure that a patient accepts as therapy but that has no known mechanism of action other than a patient’s belief in its value. Comparison against a placebo is considered the most powerful tool in evaluating the isolated effects of a procedure or treatment on a patient and his or her disease process. The use of a placebo as the control arm of a study is allowed under any of the following circumstances [18]:
- There is no standard treatment;
- New evidence has cast doubt on the standard treatment’s benefits or definitively shown it to be no more beneficial than placebo;
- The standard treatment is unavailable due to high cost or short supply;
- The standard treatment has not worked well for a specific patient population;
- An add-on to the standard treatment is being considered; or
- Adequately informed patients have consented to forgo the standard therapy for a minor ailment, like the common cold.
Sham Surgery
The term “sham surgery” is often used when a placebo procedure is used in a surgical trial. “Sham” derives from a Middle English variant of “shame” [19]. As the word suggests, sham surgery has historically been ethically controversial. There is an essential ethical requirement that the sham surgery must pose less risk to subjects than the procedure being tested, which eliminates certain groups from participation in sham-controlled surgical studies, namely, the critically ill, the acutely traumatized, and patients whose conditions can be successfully resolved with a proven safe and effective procedure. Likewise, sham surgery in clinical research should not be confused with sham surgery in clinical care, where it has no legitimate or ethically supportable role, even when no effective therapeutic modality is available [19].
Sham surgery is considered acceptable in a clinical trial when:
- It is unclear whether a procedure offers benefits above the placebo effect, which includes benefits due to the “experience of surgery” and the postoperative care regimen;
- The risks are reduced as far as possible in the sham surgery arm without compromising trial design; or
- There is no treatment that has been shown to be better than the standard therapy. Despite these accepted indications, there has been considerable debate in the literature about the ethical acceptability of using placebos in surgical research.
How can a sham operation bring about improvement in a patient’s clinical condition that mimics a true therapeutic intervention? Patients may experience benefits from the hospitalization, better pain management, ancillary treatment, and the more active sympathy that surgery elicits from all caregivers. These benefits have been shown to include improvement in pain and quality-of-life measurements [20]. These justifications are stronger when there is no clear physiological basis for why a given surgical procedure might work, as in the Mosely trial.
Sham surgery differs from use of medical placebos in several ways—the level of risk being the most obvious one. Subjects who receive placebo medications are receiving a substance with no known medical benefit, but also no risks or side effects. In contrast, sham surgeries involve risk, pain, and deception. Any sham surgery carries the risks—such as bleeding, infection, and anesthesia complications—present in every surgical intervention. They must also cause the subject some pain and appear something like the “real” operation, or subjects will know they did not have the experimental intervention and the placebo effect will be lost. The surgeon who participates in a placebo-controlled surgical trial must also actively strive to deceive the subject. This calculated deception is the basis for the potential power of the sham operation to influence the subject’s condition [21].
Critics of sham procedures point out that the use of a procedure that could cause harm without offering a compensating physiologic benefit poses ethical problems and might violate the principle of nonmaleficence [22]. This has led renowned ethicist Ruth Macklin to conclude that “performing surgery in research subjects that has no potential of therapeutic benefit fails to minimize the risk of harm” [23]. Opponents of sham operations argue that, if an intervention of proven effectiveness already exists, and if there is genuine disagreement among medical experts as to whether the new intervention is equally or more effective, then the new intervention must be compared against the established treatment rather than a placebo. If no such established intervention exists, the study intervention may be compared only against a benign placebo [17].
Miller believes that the sham arthroscopic surgeries reported by Moseley and colleagues were warranted because the procedures were relatively innocuous and the research had such clear value for evaluating a common intervention used by over half a million patients a year with total health care costs of some $3 billion [24]. The osteoarthritis study appears to have been methodologically necessary to achieve valid results and was conducted in accordance with ethical guidelines.
An implicit assumption underlies much of the debate over sham surgery—that rational people would not want to have their symptoms relieved by a procedure that draws upon their state of mind instead of succeeding through some intrinsic physiologic effect of the intervention itself. One may ask why we shouldn’t learn how to use or enhance these beneficial psychological effects rather than eliminate them. It is not surprising that half of patients reported that the intervention diminished their pain, because according to Moseley et al., “Postoperative care was delivered according to a protocol specifying that all patients should receive the same walking aids, graduated exercise program, and analgesics” [13].
Should our goal be to reduce or to stimulate placebo effects? One may argue that it is unethical not to use sham surgery to thoroughly evaluate a surgical procedure before it is introduced into clinical practice. Whatever we decide, it is at least clear that a variety of sham surgeries might be of significant value to both patients and medical science [25].
Case Summary and Ethical Considerations
What should the physician do if a patient in pain who is a good candidate for the intervention comes in and asks for the procedure, saying that he knows that the operation helps only about half the time? On the one hand, a formerly accepted but challenged article leads Dr. Janus to think that the surgery is not warranted or at best may be successful due to a placebo effect. On the other hand, he has provided arthroscopic surgical care to other patients whose conditions resemble Mr. Marcus’s with apparent success. The patient has requested the treatment and the physician believes that it can be safely done with a beneficial outcome. What is the physician to do?
At the office visit, a careful history and examination with appropriate radiographic studies should be performed. After a diagnosis is made, the physician should consider both noninvasive and invasive options. Conservative (nonoperative) treatment should be considered initially, followed by more invasive options if they do not bring about improvement. The concerns and goals of the patient need to be explored in the context of current evidence and the ethical imperative to minimize harm. Although one can argue that performing the surgery to achieve the placebo effect places the patient at risk for questionable gains, one can also argue that the benefits outweigh the risks. In spite of this, the surgeon should not embark on a treatment that clearly could harm the patient. Open dialogue will be necessary between patients and physicians to ensure that cost considerations are balanced with safety, ethical principles, and the needs of the patient.
As we go forward, decisions like these will continue to be an issue for patients and physicians. Ethical decisions will be pressured by cost considerations and value-driven health care. In the end, physicians may not be allowed to perform certain procedures if treatment choices are dictated by third-party payers despite an ethically sound approach by the treating physician and an informed patient.
References
- Owings M, Kozak J. Ambulatory and inpatient procedures in the United States, 1996. Vital Health Stat. 1998;13(139):1-119.
- Potts A, Harrast JJ, Harner CD, Miniaci A, Jones MH. Practice patterns for arthroscopy of osteoarthritis of the knee in the United States. Am J Sports Med. 2012;40(6):1247-1251.
- Kim S, Bosque J, Meehan JP, Jamali A, Marder R. Increase in outpatient knee arthroscopy in the United States: a comparison of National Surveys of Ambulatory Surgery, 1996 and 2006. J Bone Joint Surg Am. 2011;93(11):994-1000.
-
Baumgaertner MR, Cannon WD Jr, Vittore JM, Schmidt ES, Maurer RC. Arthroscopic debridement of the arthritic knee. Clin Orthop Relat Res. 1990;(253):197-202.
- Bert JM, Maschka K. The arthroscopic treatment of unicompartmental gonarthrosis: a five-year follow-up study of abrasion arthroplasty plus arthroscopic debridement and arthroscopic debridement alone. Arthroscopy. 1989;5(1):25-32.
- Gross DE, Brenner SL, Esformes I, Gross ML. Arthroscopic treatment of degenerative joint disease of the knee. Orthopedics. 1991;14(12):1317-1321.
-
Jackson RW, Silver R, Marans H. Arthroscopic treatment of degenerative joint disease. Arthroscopy. 1986;2:114.
- Rand JA. Role of arthroscopy in osteoarthritis of the knee. Arthroscopy. 1991;7(4):358-363.
-
Richards RN Jr, Lonergan RP. Arthroscopic surgery for relief of pain in the osteoarthritic knee. Orthopedics. 1984;7:1705-1707.
-
Jennings JE. Arthroscopic debridement as an alternative to total knee replacement. Arthroscopy. 1986;2:123-124.
-
McLaren AC, Blokker CP, Fowler PJ, Roth JN, Rock MG. Arthroscopic debridement of the knee for osteoarthritis. Can J Surg. 1991;34:595-598.
- Ogilvie-Harris DJ, Fitsialos DP. Arthroscopic management of the degenerative knee. Arthroscopy. 1991;7(2):151-157.
- Moseley JB, O’Malley K, Petersen NJ, et al. A controlled trial of arthroscopic surgery for osteoarthritis of the knee. N Engl J Med. 2002;347(2):81-88.
- Chang RW, Falconer J, Stulberg SD, Arnold WJ, Manheim LM, Dyer AR. A randomized, controlled trial of arthroscopic surgery versus closed needle joint lavage for patients with osteoarthritis of the knee. Arthritis Rheum. 1993;36(3):289-296.
-
Salisbury RB, Nottage WM, Gardner V. The effect of alignment on results in arthroscopic debridement of the degenerative knee. Clin Orthop Relat Res. 1985;(198):268-272.
-
Sprague NF III. Arthroscopic debridement for degenerative knee joint disease. Clin Orthop. 1981;160:118-123.
- London A, Kadane J. Placebos that harm: sham surgery controls in clinical trials. Stat Methods Med Res. 2002;11(5):413-427.
- McDonald P, Kulkarni A, Farrokhyar F, Bhandari M. Ethical issues in surgical research. Can J Surg. 2010;53(2):133-136.
- Jones J, McCullough L, Richman B. The ethics of sham surgery in research. J Vasc Surg. 2003;37(2):482-483.
-
Boyle K, Batzer F. Is a placebo-controlled surgical trial an oxymoron? J Minim Invasive Gynecol. 2007;14(3):278-283.
- Angelos P. Sham surgery in research: a surgeon’s view. Am J Bioeth. 2003;3(4):65-66.
- Tenery R, Rakatansky H, Riddick FA Jr, et al. Surgical “placebo” controls. Ann Surg. 2002;235(2):303-307.
- Macklin R. The ethical problems with sham surgery in clinical research. N Engl J Med. 1999;341(13):992-996.
- Miller FG. Sham surgery: an ethical analysis. Am J Bioeth. 2003;3(4):41-48.
-
Stock G. If the goal is relief, what’s wrong with a placebo? Am J Bioeth. 2003;3(4):53-54.