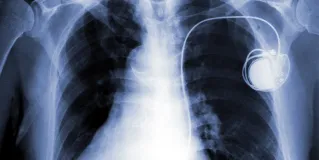
Implantable Material and Device Regulation
About 10% of Americans will have a device implanted into their bodies during their lifetimes. Fewer than 0.5% of these devices, however, are likely to have been tested in rigorous clinical trials generally regarded as standard by US regulators. Despite looser regulation of materials and devices than for pharmaceuticals, clinician-investigators and the Food and Drug Administration are obligated to balance patient-subjects’ safety with demand for patients’ timely access to technologies and interventions that might improve or extend their lives. This issue investigates that tension.