Case
Mrs. Brown, who is 35 years old, has a BMI of 37. Her father struggled with diabetes mellitus type 2 for 30 years. She was his sole caretaker, nursing him through complications of peripheral neuropathy and helping him to complete his tasks of daily living after a leg amputation. Recently, he went into renal failure and died. Mrs. Brown also has a 45-year-old brother and several other first-degree relatives who have diabetes type 2 and are insulin dependent. Mrs. Brown confided her worries to her physician and was referred to a bariatric surgeon for a consultation. She says that she has worked with nutritionists and tried to exercise more, but her efforts have not been successful over the long term. Determined to avoid becoming a diabetic, she would like to have bariatric surgery. The surgery cured her friend’s diabetes. With a BMI of 37 and no obesity-related diseases, Mrs. Brown does not qualify as a candidate for the surgery under the current guidelines. But were she either to gain weight (raising her BMI to 40) or develop diabetes (a condition which would lower the recommended BMI to 35), she would qualify for the intervention. She understands that insurance is not likely to cover the procedure, but money is not an obstacle. What should the consulting surgeon say to Mrs. Brown?
Commentary 1
The incidence of type 2 diabetes mellitus (DM type 2) in the U.S. is increasing at an alarming rate that appears to parallel the growing prevalence of obesity. The effectiveness of bariatric surgery in ameliorating DM type 2 has been well documented during the past 3 decades [1-3]. Although the mechanisms that induce weight loss among the various operations vary widely, any operation that results in substantial weight loss is likely to improve or resolve DM type II.
The weight criteria that determine candidacy for bariatric surgery were first established in the 1970s. In that early era, the minimum weight for considering bariatric surgery was 100 pounds above one’s so-called ideal body weight as established by standard life insurance tables [4]. In 1991, the NIH held a consensus development conference on gastrointestinal surgery for treatment of severe obesity. At the conclusion of that conference, the panel recommended that surgery could be considered for any patient with a body-mass index (BMI) equal to or greater than 40 for patients with a BMI between 35 and 40 who had medical diseases that most likely resulted from severe obesity [5]. These weight criteria—unmodified for nearly 2 decades—are still used by virtually all third-party payors who cover the costs of bariatric surgery.
Recently, several groups from abroad have published results of weight-loss surgery on patients with a BMI equal to or less than 35. One group prospectively compared outcomes after laparoscopic adjustable gastric banding (LAGB) and a diet/exercise program in patients with a BMI between 30 and 35. After 2 years weight loss, evidence of the metabolic syndrome and quality of life were significantly improved in the LAGB group compared with the nonsurgery group [6].
Prophylactic Surgery
The concept of “prophylactic surgery” is not new, and its use to avoid complications of the underlying disease has been ethically justified in a variety of areas. Until recently the strategy of repairing asymptomatic inguinal hernias to prevent incarceration was almost universally applied. Likewise, cholecystectomy is frequently recommended for asymptomatic gallstones to avoid subsequent complications. Repair of congenital atrial or ventricular septal defects in children is routinely performed to avert cardiopulmonary disease in adulthood, and incidental appendectomy to eliminate the potential for later appendicitis is still performed by many surgeons during abdominal operations for other causes. In each of these circumstances, the surgery is justified on the perceived basis of a favorable risk-to-benefit ratio.
Risks Associated with Bariatric Surgery
The perioperative risks associated with bariatric surgery have decreased substantially during the past decade. The mortality risk of all currently performed bariatric operations is less than 1 percent, ranging from perhaps 0.1 percent with LAGB to nearly 1.0 percent for biliopancreatic diversion with the duodenal switch (BPD/DS) [7-9]. The mortality rates of Roux-en-Y gastric bypass (RYGB) and the new sleeve gastrectomy (SG) fall somewhere in between [8-9]. Increased morbidity and mortality with RYGB is consistently correlated with male gender, age 50 years or older, and BMI equal to or greater than 50 [10, 11].
Prophylactic Bariatric Surgery
Assuming that Mrs. Brown in the case scenario we are asked to consider has made serious attempts at weight loss using dieting in conjunction with exercise and behavior modification, I believe it is ethical to perform bariatric surgery. The perioperative risks in a woman of Mrs. Brown’s age who has a BMI of 37 and no overt comorbidities should be very low. Conversely, the potential benefit of avoiding DM type 2, with its attendant end-organ complications, seems worthy of pursuit. Mrs. Brown’s strong family history of both obesity and DM type 2 suggests that eventual development of diabetes is likely. Moreover, in evaluating Mrs. Brown’s lifetime health, the risks associated with clinically severe obesity (defined as BMI equal to or greater than 35) cannot be ignored. The mortality risk at her current weight is more than double that of a woman of the same age with normal weight [12]. Life table models suggest that a 40-year-old woman with a BMI of 40 will live about 4 years less than her normal-weight counterpart [13]. Moreover, virtually all morbidly obese patients will develop obesity-related comorbidities over time. Our group reported presence of at least one obesity-related comorbidity in 95 percent of our bariatric surgery patients who were 45 years or older [14].
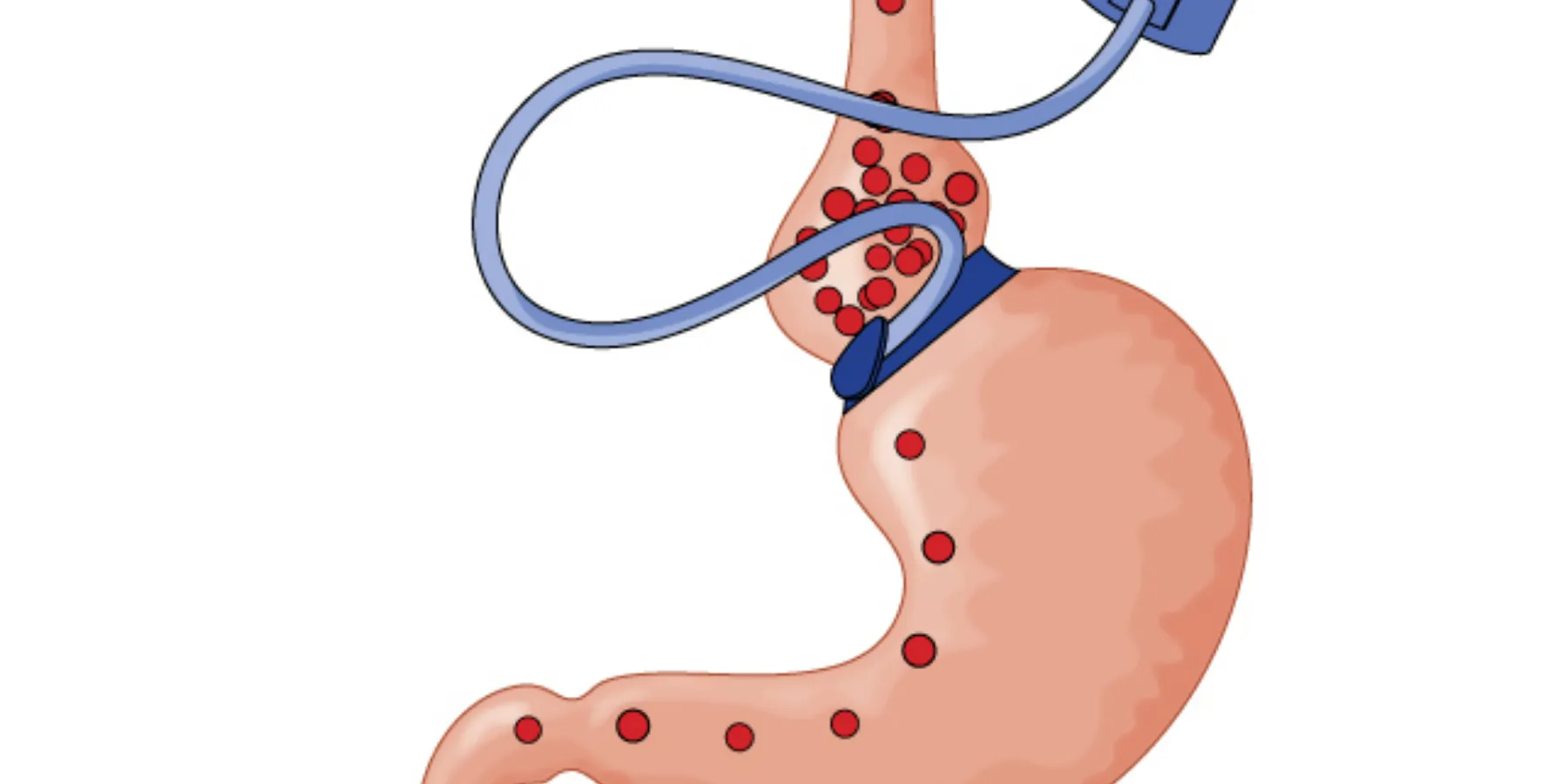
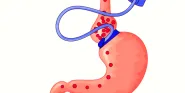
Selection of the most appropriate operation for Mrs. Brown requires a detailed discussion with her bariatric surgeon. Most bariatric surgery patients have a strong preference for a specific operation prior to their initial surgical consultation. Frequently these preferences are based entirely upon anecdotal information gleaned from other bariatric surgery patients and materials available through the internet. It is uncommon, however, for prospective patients to have a clear understanding of the risks of the various procedures or how specific operations produce weight loss. I would present both LAGB and RYGB as reasonable alternatives for Mrs. Brown. (Sleeve gastrectomy might also be considered, but the long-term results are unknown, and, although the BPD/DS provides excellent long-term weight loss in a clear majority of patients, the metabolic risk seems excessive for a woman without overt comorbidities and a BMI of 37 [15].)
Because there is no anatomical rearrangement or malabsorption with LAGB, improvement of DM type 2 is directly related to postoperative weight loss. LAGB requires considerable patient compliance in terms of the adjustments involved with tightening the band. Weight loss after RYGB is greater and more rapid than with LAGB. Moreover, DM type 2 may resolve immediately after RYGB prior to substantial weight loss [2]. These benefits must be contrasted with the long-term risks of slip or device malfunction after LAGB or the potential risks of marginal ulcer and vitamin and mineral deficiency that can develop after RYGB.
In summary, there is little if any justification for waiting until Mrs. Brown gains weight to perform bariatric surgery. The available data strongly suggest that the long-term mortality risk of not having bariatric surgery in qualified patients is significantly greater than having a gastric restrictive operation during the same time interval [1, 16, 17].
References
- MacDonald KG, Long SD, Swanson MS, et al. The gastric bypass operation reduces the progression and mortality of non-insulin-dependent diabetes mellitus. J Gastrointest Surg. 1997;1(3):213-220.
- Schauer PR, Burguera B, Ikramuddin S, et al. Effect of laparoscopic Roux-en-Y gastric bypass on type 2 diabetes mellitus. Ann Surg. 2003;238(4):467-484.
- Dixon JB, O’Brien PE, Playfair J, et al. Adjustable gastric banding and conventional therapy for type 2 diabetes: a randomized controlled trial. JAMA. 2008;299(3):316-323.
-
Measurement of Overweight. Statis Bull Metrop Insur Co. 1984; 65(1):20-23.
-
Gastrointestinal surgery for severe obesity: National Institutes of Health Consensus Development Conference Statement. Am J Clin Nutr. 1992;55(2 Suppl):615S-619S.
- O’Brien PE, Dixon JB, Laurie C, et al. Treatment of mild to moderate obesity with laparoscopic adjustable gastric banding or an intensive medical program: a randomized trial. Ann Int Med. 2006;144(9):625-633.
- Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systemic review and meta-analysis. JAMA. 2004;292(14):1724-1737.
- Clinical Issues Committee of the American Society for Metabolic and Bariatric Surgery. Sleeve gastrectomy as a bariatric procedure. Surg Obes Relat Dis. 2007;3(6):573-576.
- Podnos YD, Jimenez JC, Wilson SF, et al. Complications after laparoscopic gastric bypass: a review of 3464 cases. Arch Surg. 2003;138(9):957-961.
- DeMaria EJ, Portenier D, Wolfe L. Obesity surgery mortality risk score: proposal for a clinically useful score to predict mortality risk in patients undergoing gastric bypass. Surg Obes Relat Dis. 2007;3(2):134-140.
- Livingston EH, Huerta H, Arthur D, et al. Male gender is a predictor of morbidity and age a predictor of mortality for patients undergoing gastric bypass surgery. Ann Surg. 2002;236(5):576-582.
- Lew, EA. Mortality and weight: insured lives and the American Cancer Society studies. Ann Int Med. 1985;103(6 Pt 2):1024-1029.
- Fontaine KR, Redden DT, Wang C, et al. Years of life lost due to obesity. JAMA. 2003;289(2):187-193.
- Brolin RE, Kenler HA, Gorman RC, Cody RP. The dilemma of outcome assessment after operation for morbid obesity. Surg. 1989;105(3):337-346.
- Lagace M, Marceau P, Marceau S, et al. Bilopancreatic diversion with a new type of gastrectomy: some previous conclusions revisited. Obes Surg. 1995;5(4):411-418.
- Sjostrom L, Narbro K, Sjostrom CD, et al. Swedish Obese Subjects Study. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357(8):741-752.
- Adams TD, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;357(8):753-761.