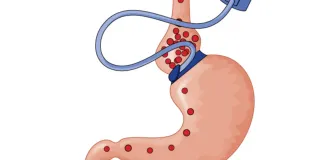
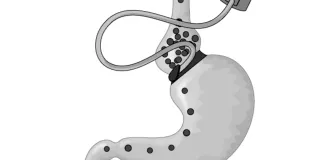
Ethics and Innovation in Surgery
Innovations in surgical techniques and devices come about in several ways. There is design, development, and controlled research, just as there is with new drugs. Often, however, in the course of routine surgery, an emergency occurs that requires lifesaving improvisation. Or surgeons may introduce planned variations to accepted techniques to improve results. Finally, surgery that has been accepted and validated for one use may be applied in novel and experimental (i.e., “off-label”) ways. February contributors explore the ethical concerns about informed consent and exposure to harm posed by all forms of innovative surgery.