Doctors’ desire to innovate is fueled by the moments when patients look to them for solutions and they have none to give. Quick adoption of new technologies, however, can be a double-edged sword. The potential to help future patients must be weighed against the risks of harming those who currently seek care. Medical innovation pits beneficence and nonmaleficence against one another in the name of progress, creating tension between physicians’ most fundamental values.
During my surgery rotation in the third year of medical school, I became acutely aware of how subjective the decision to operate may be. Many times, the decision to enter the OR is equivalent to choosing between life and death. I observed several instances in which surgeons who wanted to withdraw care were at odds with those more inclined to operate. Where some saw operations that prolonged agony, others saw opportunities for tiny victories that offered patients a little more time. There is no single formula to compare a patient's quality of life to the suffering that may possibly be caused by surgery. Whether we adopt or eschew new technologies, lives always hang in the balance.
The term “innovation” suggests advancement, just as “evolution” connotes progress toward something better. Scientific innovations are desirable because they create new possibilities and offer better performance; in the marketplace, consumers choose new over old. But the tendency to conflate “newest” with “best” can be dangerous in health care settings. This issue of Virtual Mentor explores the possibilities and perils of developing, testing, and embracing new procedures, devices, and techniques in the surgical suite. This month’s contributors touch on innovations in surgery at every phase of their development, from design and research to FDA approval and postmarket adoption into clinical practice.
The first phase of innovation for surgical devices is their design and development. During this stage, business entrepreneurs and scientists unite to identify unsolved problems and develop widely applicable solutions. Kevin Z. Chao, Daniel J. Riskin, and Thomas M. Krummel explain how the Stanford Biodesign program teaches innovation as its students work to create new devices. A formal education in this field can encourage responsible innovation. The lengths to which device developers and their employees must go to ensure responsible use of their products are controversial. This month’s health law article by Kristin E. Schleiter considers the phenomenon in which manufacturer’s representatives join surgeons in the OR to ensure that devices function properly and are correctly employed. Court decisions have clarified the point at which device makers cease to be held liable for the performance of surgeons who use their approved technologies.
In research on innovative surgeries, patient data is used to develop knowledge and new techniques for future application to patients with similar conditions [1]. Safeguards that protect patients from harm during research include the informed consent process and oversight by institutional review boards (IRBs). Research differs from other paths to innovation in surgery because IRB approval is required for trial protocols, subject recruitment methods, informed consent, and so on. A case commentary by Robert Sade asks whether surgeons in clinical trials have an obligation to provide information above and beyond what is on the IRB-approved consent form.
Innovation in surgery also occurs outside the research process. As Joseph Fins has written, therapeutic, validated surgery has the potential to become innovative, and perhaps experimental, depending on the situation [2]. Unplanned experimental innovations happen during emergencies, when surgeons follow protocol until lifesaving improvisation is required [3]. Informed consent is not typically feasible, but is considered implied because the alternative is immediate death or serious morbidity. Non-experimental innovations are the result of planned variations on accepted techniques. The changes predictably improve results but are sufficiently minor that consenting patients need not acknowledge them.
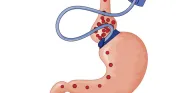
Somewhere between clinical improvisation and research lie instances in which proven protocols gain off-label use. When off-label use becomes widespread, the new application may re-enter the validation process. Caitlin Weber’s piece examines the controversies surrounding FDA-approved products that are used to achieve new endpoints or applied to untested subsets of patients, such as children. Weber demonstrates that guidelines have yet to be clearly established for re-evaluation of procedures that become innovative when applied in a new context. The ethics of performing surgery off-label for new indications outside of the approved patient population is debated in the second clinical case. Commentaries by Robert E. Brolin, Angelique M. Reitsma, and Bruce Schirmer take different stances on the appropriateness of a new application for an approved and time-tested surgery: bariatric surgery as a preventive measure against type 2 diabetes.
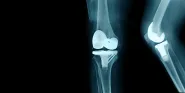
Aspects of surgical protocol over which surgeons typically exercise autonomy—including favoring newer device brands over others—can influence a procedure’s outcome. Device- or surgeon-specific variability within a given type of procedure is common but difficult to regulate. One possible way to oversee device selection without sacrificing physician autonomy is the model provided by national joint registries. As Fabian von Knoch, Anthony Marchie, and Henrik Malchau note, some devices are found to be defective or to cause complications for a particular group in the postmarket stage. Patients in countries with national registries greatly benefit from comprehensive tracking of success rates for new joint implants. In his case commentary, Charles Rosen strongly advocates that surgeons disclose the experimental status of new devices and give information about their postmarket success rates during the informed consent process. The clinical pearl, by Allen Carl, delves into the spine stabilization technology featured in Rosen’s commentary, explaining how the latest surgical techniques were developed and why they remain controversial.
Surgical innovation does not always involve new surgeries or implants; it may also refer to changes in the OR setting. The standardization of operating team procedures might, at first, be considered conservative by surgeons accustomed to a culture of autonomy and individualism. Julie Ann Freischlag points out that, in fact, OR safety measures are an innovative way of improving patient outcomes. As Ankur Aggarwal’s piece on the history of surgery explains, similar safety-oriented changes were able to improve medical doctors’ perception of surgery over the course of human civilization. Decried as dangerous butchery, last-ditch attempts to save lives with radical surgery were assaults on patients and “mutilation and suffering [were] caused by too late and hopeless operations” [4]. Advances in sterile technique and anesthesiology elevated surgery to a respected and trusted field with a high success rate.
Innovation in surgery has its costs to patients and surgeons as well. Thomas Starzl, one of the fathers of transplant surgery, notes that “hardly a transplant surgeons in that era [of the 1960s] escaped infection [with hepatitis]. My chief research technician...died from hepatitis and so did many others. Eventually, it was proved that a hepatitis reservoir existed in the transplant wards and clinics.” Starzl, too, was infected with hepatitis [5]. His journey to success entailed great personal sacrifice, yet in his memoirs he insists that the benefits far outweighed the risks of the innovative procedures he performed. Medical advancements also have costs to insurance companies and the health care system. This month’s medicine and society piece, by Joseph J. Fins, weighs the costs, literal and figurative, of controversial central thalamic deep brain stimulation (DBS). Fins persuasively argues that the advantages of DBS are substantial—not only in terms of therapeutic benefit, but also, surprisingly, economically.
Despite the promise of innovative surgery, its potential to do harm with untested, unsafe, or inappropriate procedures remains. If undertaken responsibly, innovation can promote the best interests of both the individual patient and society as a whole. Great ideas may spring from creativity, but it is only when coupled with vigilant attention to patient safety that they lay the groundwork for great progress.
References
-
Jones JW. The surgeon’s autonomy: defining limits in therapeutic decision making. In: Reitsma AM, Moreno J, eds. Ethical Guidelines for Innovative Surgery. Hagerstown, MD: University Publishing Group; 2006:74-92.
- Fins JJ. Surgical innovation and the ethical dilemmas: precautions and proximity. Cleve Clin J Med. 2008;75(Suppl 6):S7-S12.
-
McCullough LB. Standard of care, innovation, and research in surgery: a problem in research ethics or in professional medical ethics? In: Reitsma AM, Moreno J, eds. Ethical Guidelines for Innovative Surgery. Hagerstown, MD: University Publishing Group; 2006:53-73.
- Gage H. Some abuses in surgical practice. Boston Med Surg J. 1913;1691-7.
- Starzl TE. In a small Iowa town. Transplant Proc. 1988;20(1 Suppl 1):12-17.