Abstract
Economics is the primary discipline used to understand supply chain design, scale-up, and management. For example, antibiotics can be compared to other forms of “tragedy of the commons,” whereby a common good (effective treatment of infections) is jeopardized by individual consumption and lack of community oversight and stewardship. While economic analysis can explain innovation decline in terms of market failure, one pitfall of an early-stage focus on research and development is a failure to challenge the discovery narrative. Ethics also has a distinct place in helping us envision alternatives to what markets can produce. This article advances a more contextualized view of how science and technology policy has shaped antibiotic supply chains over many years, emphasizing how shifting the story we tell about past successes is central to securing a reliable antibiotic supply chain in the future.
Effectiveness Paradox
Antimicrobials are treatments for microbial infections caused by bacteria, viruses, and fungi. Antibiotics are medicines especially used to treat bacterial infections. Penicillin was the first antibiotic and effective treatment developed for bacterial infections encompassing pneumonia, gonorrhea, and rheumatic fever.1 Penicillin’s reduction in human suffering is neither qualitatively nor quantitatively simple to capture, but the medicine has saved millions of lives and improved human life expectancy for many. However, antibiotics present an “effectiveness paradox”: the more they are used, the less effective they become. Repeat exposure of bacteria to an antibiotic can generate the conditions that select for resistance, or the capacity of colonies to survive despite treatment. One recent study estimated that over 1.27 million deaths in 2019 were attributable to bacterial antimicrobial-resistant infections.2 As antibiotic resistance is increasing globally, so, too, is demand for last-resort medicines that can effectively treat resistant infections.3 The effectiveness paradox can be compared to other forms of “tragedy of the commons,” whereby a common good (eg, effective treatment of infections) can be jeopardized by individual consumption.4 However, as Hardin recognized, ethics has a distinct place in helping us envision alternatives to the tragedy of the commons.5 This article advances a more contextualized view of how value-driven science and technology policy has shaped antibiotic supply chains over the years,6 emphasizing how the story we tell about past success is central to securing access to antibiotics in the future.
A Story of Antimicrobial Innovation
The sheer magnitude of lives saved by antibiotics is a staggering public health, medical, and humanitarian achievement. It is unsurprising, then, that the advent of antibiotics is among the historical developments that have the hallmarks of heroic stories. The discovery narrative is linear, simple, and marked by regular innovations of distinctive scientific personalities. Commonly depicted along a timeline, the 1950s to 1970s period of antibiotic development is often referred to as the “golden age” of antibiotics (see Figure).7,8
Figure. Dominant Narrative of Antibiotic Discovery
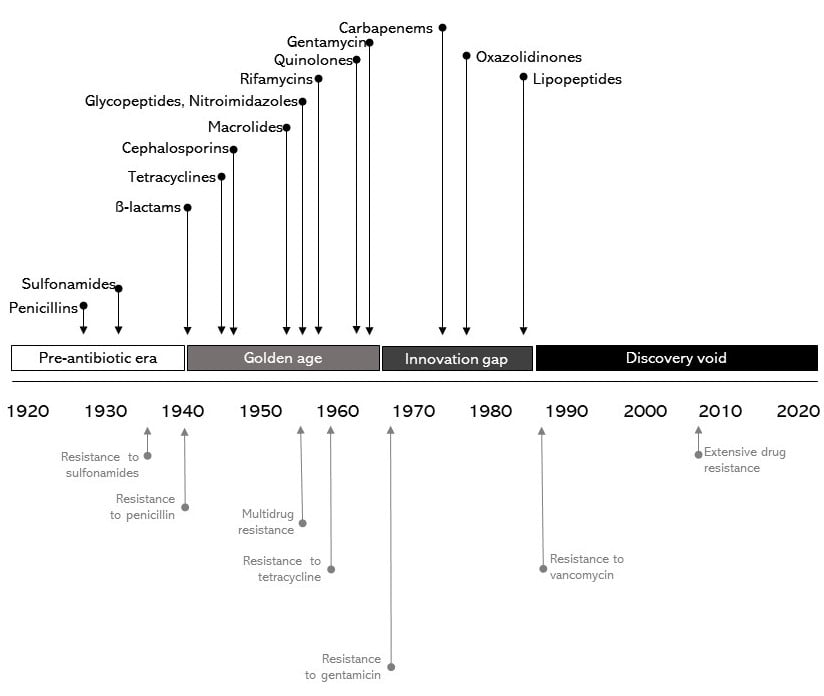
Data sources: Iskandar K, Murugaiyan J, Hammoudi Halat D, et al7; Silver LL8; Ventola CL9; National Research Council10; Davies J, Davies D11; Rahman MM, Alam Tumpa MA, Zehravi M, et al.12
The discovery narrative is in keeping with Paul De Kruif’s depiction of big scientific personalities as primary enactors of scientific achievement, which he chronicled in his 1926 influential book, Microbe Hunters.13 De Kruif focused on microbiologists of the 19th century, and his account is one of steady progress: “it is sure as the sun following the dawn of tomorrow, that the high deeds of microbe hunters have not come to an end; there will be others to fashion magic bullets.”13 Independently of who merits credit for the achievement of identifying penicillin’s medical utility and refining its production, Alexander Fleming’s ability to fit within the discovery narrative might partly explain the messy media storm that characterized him as the sole scientific genius who revolutionized medicine with penicillin.14,15 In contrast, Howard Florey’s more reserved personality and his team’s collective efforts to purify and test the effectiveness of penicillin at the University of Oxford garnered much less public attention and received delayed recognition.15
Diverging from De Kruif’s vision of steady progress, contemporary drug development is frequently depicted as an era of a “discovery void” following an “innovation gap” in which new antibiotic drug development petered out in the 1980s and 1990s (see Figure). The same timeline of bygone halcyon days followed by a fallow period has been presented across popular media, pharmacology, microbiology, and policy.7,8,9,10,11,16 The failure of the 21st century to live up to the promise of progress clashes with a protagonist-driven account of how scientific success occurs. For antibiotics, the oft-unexamined link between discovery and scientific heroism is so tight that, for the last decade, the phase following the “lean years” on timelines has been depicted as one of “disenchantment,”—a future that is oddly anachronistic, given that it is often explicitly depicted as a post-antibiotic return to the 1800s and the time of Semmelweis, a physician from 200 years ago with no antibiotic armamentarium except his (widely ignored) advocacy of hand hygiene.11,12 It is notable that within the discovery narrative, there is little examination of how scientific heroism accords with a profit motive. (See Supplementary Appendix on economic concepts related to antibiotic resistance.) This lacuna in the dominant narrative of penicillin is especially striking, as patent debates marked disagreements within the scientific community from the very beginning.17 Timelines like those in the Figure demonstrate how discovery narratives continue to shape popular understanding of how science progresses. Gaps in our understanding of what drives functional antibiotic supply chains are partly due to this tendency to decouple the history of science from its social and political context.
Some turn to economics to account for the contrast between antibiotics’ profound contributions to human well-being and the current period of innovation stagnation, seeking a solution to the tragedy of the commons in a market-driven pricing model.18 It has long been widely recognized that markets confront serious limitations in their ability to supply medical services efficiently.19 Antibiotic market failures that lead to the detriment of social well-being are depicted as “deviations” from ideal economic market dynamics that concern only 2 parties (producers and consumers) and a range of stipulated conditions that enable markets to efficiently meet consumer needs. More specifically, economic analyses emphasize how, since the 1970s, the lack of landmark antibiotic discoveries is due to sudden or newly emerging market failures such as lack of large profit margins (when compared to treatment for chronic diseases), price deviations from social value, and stewardship practices that undermine sales by volume.20 A perception that market incentives for antibiotics are misaligned has led to developments such as CARB-X (Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator), a nonprofit organization that funds preclinical and early phase novel antimicrobial research.21
Economics does provide one way to understand the market failures that contribute to a paucity of innovation and can be consonant with ethical perspectives.22 Antibiotic resistance is a prime example of an externality that contributes to market failure: a cost borne by society as a whole as infections become more difficult to treat. Industrialized agriculture pollution of waterways that results in antibiotic resistance is another form of externality.23 However, lumping together many structural failures under the label of “externalities” can conflate the value of antibiotics as prevention and treatment, obfuscate the responsibilities of states to protect public health, and evade identification of social structures that supply goods in ways that go beyond consumer satisfaction and efficiency (eg, equitably and sustainably). Other examples of market failure in antibiotic supply include monopoly (oligopoly) power of biopharmaceutical companies, which is sustained by high up-front innovation costs and control over manufacturing processes that leads to noncompetitive drug pricing and inadequate geographic dispersion of production capacity and supply chains.
In the next section, I explore how redirecting attention to science and technology policy provides a more comprehensive account of our past than economic explanations of market failure consonant with the discovery narrative’s focus on early-stage antibiotic research and development. I also discuss obstacles to policy change and recommended policies for moving forward.
Early Antimicrobial Production and Distribution
It was not only novel discoveries but also innovative approaches to science, technology, and health policy that rendered penicillin effective, available, and accessible both during and after the Second World War. For a short but incredibly intense period in the 1940s, the US government scaled penicillin production by modifying policies on trade secrets, property rights, antitrust regulations, and drug licensure.5,13,14 The US War Production Board (WPD) broke down the barriers of trade secrets by creating consortia of private companies, academic partners, and government agencies whose members were incentivized to share and develop industry-wide best practices for antibiotic quality and scalable production. Moreover, in contrast to the narrative of scientific heroism, it was highly collaborative cross-industry and multinational structures that led to rapid innovation and scaling up of manufacturing.5 This section provides an overview of how functional antibiotic supply for some populations was previously achieved through strategic national objectives combined with shifts in domestic and global policy encompassing science, trade, and humanitarianism.
Strategic national objectives. During the Second World War, the US government’s compelling interest was to prevent and treat infections of Armed Services personnel on the front line. The WPD consortium increased penicillin supply in part by creatively utilizing American farmers’ know-how and existing resources. For example, an agricultural research laboratory in Peoria, Illinois, helped adapt deep fermentation processes using corn-steep liquor to increase the penicillin content in each production batch.24 Meanwhile, in the United Kingdom, penicillin was such a precious resource that its use was restricted to objectives integral to the war effort. In 1941, Florey himself provided some doses to veterinarians addressing mastitis infections in cows; dairy farms were crucial to a populace whose diets were severely limited by international shortages.25
Domestic trade policy. Notably, intellectual property policies were also rearranged to support domestic penicillin development and manufacturing scale-up. Scientists in the Oxford group disagreed about the wisdom and ethics of obtaining a patent, including about whether products as opposed to processes could be considered intellectual property. Such disagreements are especially pertinent to bioethics, as it was an ethical obligation to serve humanity that shaped Florey’s decision not to patent the Oxford team’s process for producing penicillin.13,14 Meanwhile, most US process patents were held by the US Department of Agriculture and widely licensed without royalties. Quinn contends that it was the absence of product patents that enabled commercial pharmaceutical companies to create novel reciprocal licensing arrangements, engage collaboratively in ways that were far superior to competitive research and development, and share information more effectively.5 With scientific cooperation surreptitiously hidden from Nazi occupying forces, a distinct Netherlands research group refined its own process. After the war, the group’s separate patent led to both more supply and lower prices.13
Global trade policy. During the postwar era, antibiotic availability was driven by other global policy shifts that sought to recognize the distinctive global value of antibiotics. US intellectual property arrangements may have supported scalability to meet needs within the Global North, but access in the Global South still lagged. The 1970 Indian Patents Act reshaped drug manufacturing globally, in part by abolishing product-based drug patents, enabling generic versions of drugs to be produced through reverse engineering of pharmaceuticals in India.26 Antibiotics have also been at the center of determining international implications of the rule of law. For example, India, Iran, and the Philippines filed suit against Pfizer for violation of the Sherman Act by establishing monopoly practices. In 1978, in Pfizer, Inc v Government of India, the US Supreme Court recognized the status of sovereign nations to sue under US domestic law.27 India’s subsequent rapid development of pharmaceutical manufacturing, combined with a US regulatory abbreviated new drug application process in the 1980s, allowed Indian manufacturers to avoid repeating clinical trials or marketing comparable generics in the United States, resulting in India becoming a current leader in world antibiotic manufacturing and the United States becoming the largest importer of their antibiotic exports.26
Global health policy. Because antibiotics are lifesaving, ensuring access to them has been a high priority in global health policy. However, global access to antibiotics is highly variable and fragile,27,28,29 both with and without a prescription.30 The World Health Organization (WHO) included antibiotics on its essential medicines list (EML) for the first time in 1977.31 Although the WHO definition of essential medicines and its processes for listing medicines has changed over time, by 2002 the EML prioritized infectious disease health needs and articulated adequate antibiotic supply as a criterion of functional national health systems.32 More recently, the WHO has proposed categorizing antibiotics on the essential medicines list as Access, Watch, or Reserve, depending on their lifesaving potential and likelihood of generating resistance.33
Many of the economic strategies suggested by a discovery void narrative rely on leveraging policy to serve economic goals. Conversely, economics can be a tool by which we ascertain how well we are achieving antibiotic clinical and stewardship goals (eg, monitoring WHO Access-Watch-Reserve antibiotics). For example, Orubu and colleagues identified 16 indicators across the antibiotic supply chain that can be used to assess national capacity to ensure population access to antibiotics and mitigate inappropriate use, in part due to dispensaries outside the control of pharmacists.34 They found that over half of the licenses for antibiotic products in Bangladesh belonged to the WHO Watch group rather than the Access group; the authors contend that the proportion of licensed WHO Watch antibiotics on the market provides one way to measure misuse of antibiotics that might be replaced by treatment options with fewer risks of producing resistance.34
Moving Forward
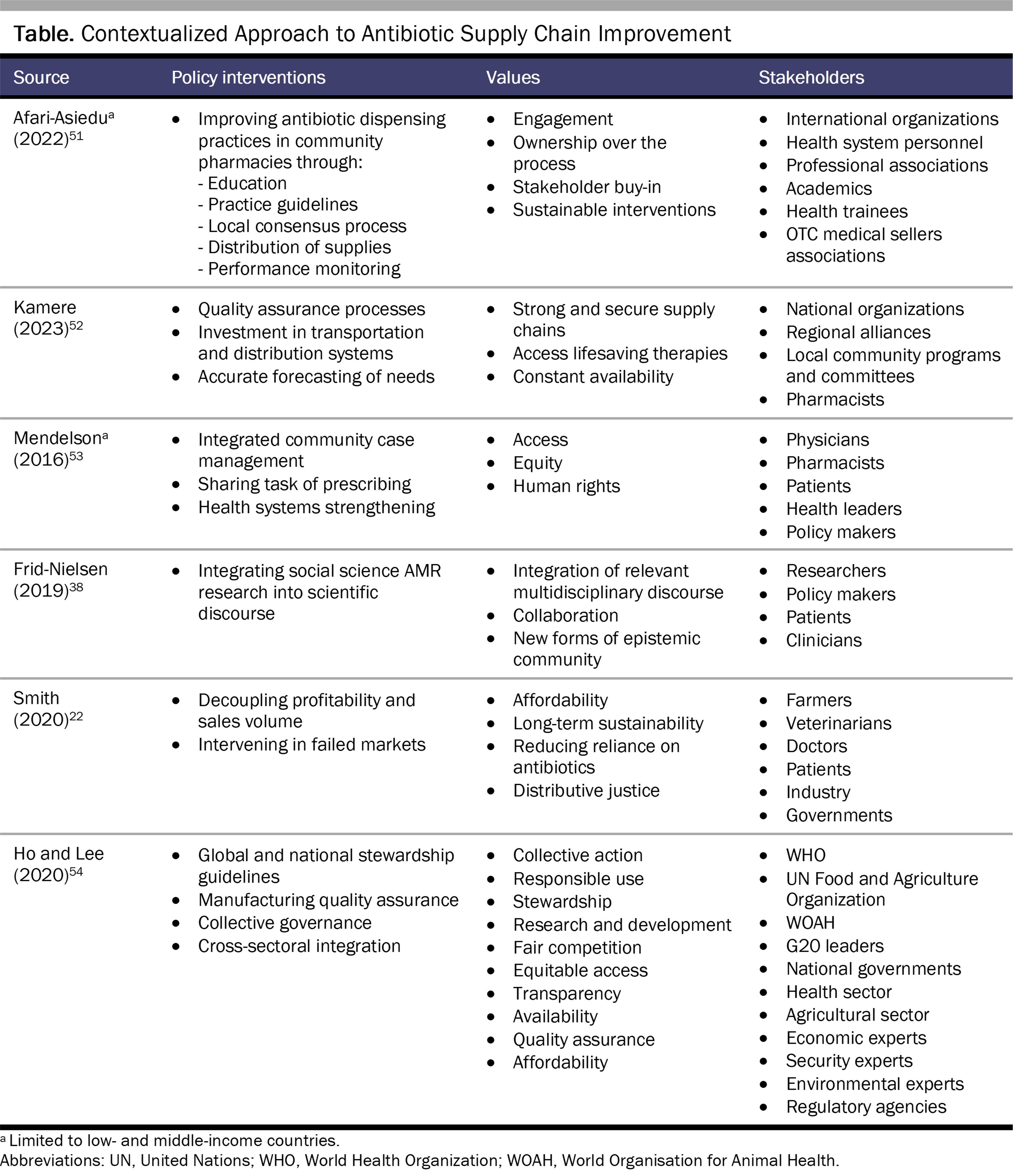
Current policy interventions for mitigating the rise of antibiotic resistance are wide-ranging, including price controls, taxation, improved surveillance, legal reform, health services infrastructure investment, public and expert educational initiatives, pharmacy guidance, and regulatory oversight of agricultural or human use.35,36,37 Bioethics and social science have also offered a variety of contributions that draw on economic, anthropological, sociological, historical, and normative approaches.25,38,39,40,41,42,43,44 These discourses share the insight that the drivers of resistance are sufficiently complex that coordinated policy solutions that cross national and geographic boundaries are needed.45,46 As the Bangladeshi study demonstrates, attending to policy and socio-behavioral dynamics of antibiotic resistance also redirects attention to the evidence base for stewarding antibiotics, including both facilitators and barriers.47,48,49 Collaborative effort could leverage multidisciplinary insights, with cultural analysis50 and ethical analysis helping to identify values reflected in policy alternatives, values-based attitudes of stakeholders, and justificatory grounds of policy change. The resources listed in the Table focus specifically on policies that can improve antibiotic supply and distribution. These resources provide initial insight into formulating multidisciplinary research questions that can advance more contextualized approaches to antibiotic supply chain policy.
Conclusion
In sum, the “golden age” of antibiotics is arguably a sociopolitical story, one that recapitulates the tendency to nostalgically view the 1950s through 1970s as a bygone heyday of the United States’ rise to global dominance, including through strategic advancement of science and technology. The discovery narrative, however, fails to explicate how the benefits of antibiotics were and continue to be accrued by some groups while excluding others. Governments have always intervened in antibiotic production, and therefore the “innovation gap” does not reflect a novel state of market failure in antibiotic supply chains. Rather, the benefits and harms of antibiotic usage extend well beyond the innovation stage. Relinquishing the dominant, ahistorical discovery narrative is the first step to redirecting our analyses appropriately: toward questioning how the rise of antibiotics resistance has failed to generate the political will necessary to propel science and technology policies that prioritize access, equity, and sustainability.
References
-
Allen HB, Hossain C, Abidi N, et al. Penicillin: the old/new wonder drug. Adv Tech Biol Med. 2017;5(1):1000197.
-
Murray CJ, Ikuta KS, Sharara F, et al; Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399(10325):629-655.
- Klein EY, Milkowska-Shibata M, Tseng KK, et al. Assessment of WHO antibiotic consumption and access targets in 76 countries, 2000-15: an analysis of pharmaceutical sales data. Lancet Infect Dis. 2021;21(1):107-115.
- Hollis A, Maybarduk P. Antibiotic resistance is a tragedy of the commons that necessitates global cooperation. J Law Med Ethics. 2015;43(suppl 3):33-37.
- Hardin G. The tragedy of the commons: the population problem has no technical solution; it requires a fundamental extension in morality. Science. 1968;162(3859):1243-1248.
- Quinn R. Rethinking antibiotic research and development: World War II and the penicillin collaborative. Am J Public Health. 2013;103(3):426-434.
-
Iskandar K, Murugaiyan J, Hammoudi Halat D, et al. Antibiotic discovery and resistance: the chase and the race. Antibiotics (Basel). 2022;11(2):182.
- Silver LL. Challenges of antibacterial discovery. Clin Microbiol Rev. 2011;24(1):71-109.
- Ventola CL. The antibiotic resistance crisis: part 1: causes and threats. P T. 2015;40(4):277-283.
-
Friedman D, Alper J; National Research Council. Challenges in overcoming antibiotic resistance. In: Technological Challenges in Antibiotic Discovery and Development: A Workshop Summary. National Academies Press; 2014:chap 2.
-
Davies J, Davies D. Origins and evolution of antibiotic resistance. Microbiol Mol Biol Rev. 2010;74(3):417-433.
-
Rahman MM, Alam Tumpa MA, Zehravi M, et al. An overview of antimicrobial stewardship optimization: the use of antibiotics in humans and animals to prevent resistance. Antibiotics (Basel). 2022;11(5):667.
-
De Kruif P. Microbe Hunters. Harvest Books; 1996.
- Gaynes R. The discovery of penicillin—new insights after more than 75 years of clinical use. Emerg Infect Dis. 2017;23(5):849-853.
- Goldsworthy PD, McFarlane AC. Howard Florey, Alexander Fleming and the fairy tale of penicillin. Med J Aust. 2002;176(4):176-178.
-
Hudson AO. Antibiotics resistance is at a crisis point. The Conversation. October 29, 2021. Accessed November 28, 2023. https://theconversation.com/antibiotic-resistance-is-at-a-crisis-point-government-support-for-academia-and-big-pharma-to-find-new-drugs-could-help-defeat-superbugs-169443
- Ligon BL. Sir Howard Walter Florey—the force behind the development of penicillin. Semin Pediatr Infect Dis. 2004;15(2):109-114.
-
Roope LS, Smith RD, Pouwels KB, et al. The challenge of antimicrobial resistance: what economics can contribute. Science. 2019;364(6435):eaau4679.
- Arrow KJ. Uncertainty and the welfare economics of medical care. Am Econ Rev. 1963;53(5):941-973.
- Spellberg B, Bartlett J, Wunderink R, Gilbert DN. Novel approaches are needed to develop tomorrow’s antibacterial therapies. Am J Respir Crit Care Med. 2015;191(2):135-140.
-
Supporting great science from around the world. CARB-X. Accessed November 28, 2023. https://carb-x.org/portfolio/portfolio-companies/
-
Smith RD, Coast J. The economics of resistance through an ethical lens. In: Jamrozik E, Selgelid M, eds. Ethics and Drug Resistance: Collective Responsibility for Global Public Health. Springer; 2020:279-294.
-
Hubeny J, Harnisz M, Korzeniewska E, et al. Industrialization as a source of heavy metals and antibiotics which can enhance the antibiotic resistance in wastewater, sewage sludge and river water. PLoS One. 2021;16(6):e0252691.
- Henderson JW. The yellow brick road to penicillin: a story of serendipity. Mayo Clin Proc. 1997;72(7):683-687.
-
Kirchelle C. Pyrrhic Progress: The History of Antibiotics in Anglo-American Food Production. Rutgers University Press; 2020.
-
Bjerke L. Antibiotic geographies and access to medicines: tracing the role of India’s pharmaceutical industry in global trade. Soc Sci Med. 2022;312:115386.
-
Pfizer, Inc v Government of India, 434 US 308 (1978).
-
Saleem Z, Saeed H, Akbar Z, et al. WHO key access antibiotics price, availability and affordability in private sector pharmacies in Pakistan. Cost Eff Resour Alloc. 2021;19(1):10.
- Laing R, Waning B, Gray A, Ford N, ’t Hoen E. 25 years of the WHO essential medicines lists: progress and challenges. Lancet. 2003;361(9370):1723-1729.
- Auta A, Hadi MA, Oga E, et al. Global access to antibiotics without prescription in community pharmacies: a systematic review and meta-analysis. J Infect. 2019;78(1):8-18.
-
World Health Organization. The selection of essential drugs: report of a WHO expert committee. World Health Organization; 1977. Accessed May 31, 2023. https://iris.who.int/bitstream/handle/10665/41272/WHO_TRS_615.pdf?sequence=1&isAllowed=y
-
WHO Expert Committee on the Use of Essential Drugs. Essential medicines: WHO model list. 12th ed. World Health Organization; 2002. https://iris.who.int/bitstream/handle/10665/67335/a76618.pdf?sequence=1&isAllowed=y
-
World Health Organization. The Selection and Use of Essential Medicines: Report of the WHO Expert Committee, 2017. World Health Organization; 2017. Accessed January 26, 2024. https://iris.who.int/bitstream/handle/10665/259481/9789241210157-eng.pdf?sequence=1
- Orubu ESF, Samad MA, Rahman MT, Zaman MH, Wirtz VJ. Mapping the antimicrobial supply chain in Bangladesh: a scoping-review-based ecological assessment approach. Glob Health Sci Pract. 2021;9(3):532-547.
- Wallinga D, Smit LAM, Davis MF, Casey JA, Nachman KE. A review of the effectiveness of current US policies on antimicrobial use in meat and poultry production. Curr Environ Health Rep. 2022;9(2):339-354.
-
Rogers Van Katwyk S, Grimshaw JM, Nkangu M, et al. Government policy interventions to reduce human antimicrobial use: a systematic review and evidence map. PLoS Med. 2019;16(6):e1002819.
-
Sakeena MHF, Bennett AA, McLachlan AJ. Enhancing pharmacists’ role in developing countries to overcome the challenge of antimicrobial resistance: a narrative review. Antimicrob Resist Infect Control. 2018;7:63.
-
Frid-Nielsen SS, Rubin O, Baekkeskov E. The state of social science research on antimicrobial resistance. Soc Sci Med. 2019;242:112596.
-
Lu J, Sheldenkar A, Lwin MO. A decade of antimicrobial resistance research in social science fields: a scientometric review. Antimicrob Resist Infect Control. 2020;9(1):178.
-
Jensen CS, Nielsen SB, Fynbo L. Risking antimicrobial resistance: a One Health study of antibiotic use and its societal aspects. In: Jensen CS, Nielsen SB, Fynbo L, eds. Risking Antimicrobial Resistance. Palgrave Macmillan; 2019:1-24.
- Antoine-Moussiaux N, Janssens de Bisthoven L, Leyens S, et al. The good, the bad and the ugly: framing debates on nature in a One Health community. Sustain Sci. 2019;14(6):1729-1738.
- Jamrozik E, Heriot GS. Ethics and antibiotic resistance. Brit Med Bull. 2022;141(1):4-14.
-
Hoffman SJ, Outterson K. What will it take to address the global threat of antibiotic resistance? J Law Med Ethics. 2015;43(suppl 3):6-11.
- Littmann J, Viens AM. The ethical significance of antimicrobial resistance. Public Health Ethics. 2015;8(3):209-224.
-
Littmann J, Viens AM, Silva DS. The super-wicked problem of antimicrobial resistance. In: Jamrozik E, Selgelid M, eds. Ethics and Drug Resistance: Collective Responsibility for Global Public Health. Springer; 2020:421-443.
- Van Katwyk SR, Grimshaw JM, Hoffman SJ. Ten years of inaction on antimicrobial resistance: an environmental scan of policies in Canada from 2008 to 2018. Healthc Policy. 2020;15(4):48-62.
- Hulscher MEJL, Prins JM. Antibiotic stewardship: does it work in hospital practice? A review of the evidence base. Clin Microbiol Infect. 2017;23(11):799-805.
-
Rzewuska M, Charani E, Clarkson JE, et al; Joint Programming Initiative on Antimicrobial Resistance (JPIAMR) Working Group on Behavioural Approaches to Antibiotic Stewardship Programs. Prioritizing research areas for antibiotic stewardship programmes in hospitals: a behavioural perspective consensus paper. Clin Microbiol Infect. 2019;25(2):163-168.
- Ghiga I, Sidorchuk A, Pitchforth E, Stålsby Lundborg C, Machowska A. “If you want to go far, go together”—community-based behaviour change interventions to improve antibiotic use: a systematic review of quantitative and qualitative evidence. J Antimicrob Chemother. 2023;78(6):1344-1353.
-
Ledingham K, Hinchliffe S, Jackson M, Thomas F, Tomson G. Antibiotic resistance: using a cultural contexts of health approach to address a global health challenge. World Health Organization Regional Office for Europe; 2019. Accessed July 27, 2023. https://iris.who.int/bitstream/handle/10665/330029/9789289053945-eng.pdf?sequence=2&isAllowed=y
-
Afari-Asiedu S, Abdulai MA, Tostmann A, et al. Interventions to improve dispensing of antibiotics at the community level in low and middle income countries: a systematic review. J Glob Antimicrob Resist. 2022;29:259-274.
- Kamere N, Rutter V, Munkombwe D, et al. Supply-chain factors and antimicrobial stewardship. Bull World Health Organ. 2023;101(6):403-411.
- Mendelson M, Røttingen JA, Gopinathan U, et al. Maximising access to achieve appropriate human antimicrobial use in low-income and middle-income countries. Lancet. 2016;387(10014):188-198.
-
Ho CWL, Lee TL. Global governance of anti-microbial resistance: a legal and regulatory toolkit. In: Jamrozik E, Selgelid M, eds. Ethics and Drug Resistance: Collective Responsibility for Global Public Health. Springer; 2020:401-420.